Bone adaptation during rehabilitation represents a critical intersection of **biology**, clinical **practice**, and patient-centered **outcomes**. Understanding how skeletal tissue responds to mechanical stimuli and therapeutic interventions is essential for optimizing recovery following injury or surgery. This article explores the foundational mechanisms of bone adaptation, outlines key rehabilitation strategies, and highlights emerging technologies poised to transform patient care.
Biological Foundations of Bone Adaptation
Mechanotransduction and Cellular Players
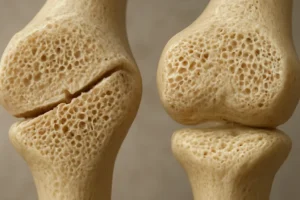
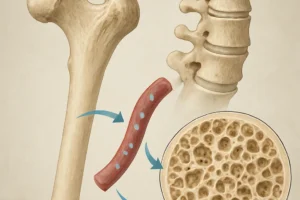
Bone tissue is a dynamic organ that constantly undergoes **remodeling** to adapt to changing mechanical environments. At the core of this process is mechanotransduction, the conversion of physical forces into biochemical signals. Specialized cells—osteocytes, osteoblasts, and osteoclasts—coordinate bone formation and resorption.
- Osteocytes: Embedded within the mineralized matrix, these cells sense mechanical loading and release signaling molecules (e.g., sclerostin) to regulate bone turnover.
- Osteoblasts: Responsible for new bone formation by depositing collagen and hydroxyapatite, they respond to signals from osteocytes and systemic hormones.
- Osteoclasts: Multinucleated cells that resorb old or damaged bone, paving the way for subsequent formation by osteoblasts.
Wolff’s Law and Adaptive Responses
According to Wolff’s Law, bone density and architecture change in response to the magnitude and direction of mechanical forces. Rehabilitation protocols leverage this principle by prescribing specific loading regimens—weight-bearing, resistance exercises, or vibration therapy—to stimulate adaptive remodeling. However, the relationship between force magnitude, frequency, and duration must be balanced to avoid stress fractures or disuse osteopenia.
Influence of Systemic Factors
Beyond mechanical stimuli, systemic factors such as hormonal milieu, nutritional status, and comorbidities play significant roles. For instance, adequate intake of vitamin D and calcium is essential for matrix mineralization, while chronic conditions like diabetes or rheumatoid arthritis can impair bone quality. Tailoring rehabilitation plans requires a holistic assessment of these variables.
Therapeutic Strategies in Rehabilitation
Progressive Weight-Bearing Protocols
Early controlled weight-bearing has become a cornerstone of bone injury rehabilitation. By gradually increasing mechanical loads, clinicians can enhance **bone** strength while minimizing the risk of reinjury. Key principles include:
- Initiation of partial weight-bearing as tolerated, often guided by pain levels and imaging findings.
- Incremental progression to full weight-bearing using assistive devices (e.g., crutches, walkers) and monitored by gait analysis.
- Integration of functional tasks, such as stair climbing or balance challenges, to promote physiologic loading directions.
Resistance Training and Muscle-Bone Interactions
Muscle contractions exert internal forces on bone, stimulating adaptive responses. Resistance training protocols—utilizing elastic bands, free weights, or pneumatic machines—target **muscle-bone interactions**. Research indicates that high-impact, multidirectional loading may yield superior bone density improvements compared to unidirectional exercises. Clinicians should consider patient-specific factors such as age, bone health baseline, and comorbidities when prescribing resistance regimens.
Adjunctive Therapies: Vibration and Electrical Stimulation
Emerging adjuncts include low-magnitude, high-frequency vibration platforms and pulsed electromagnetic fields. These modalities aim to mimic micro-mechanical strains, enhancing osteoblastic activity without imposing excessive macroscopic loads. Additionally, functional electrical stimulation can **activate** paralyzed or weakened muscles, indirectly benefiting bone health by restoring dynamic loading patterns.
Biomechanical Considerations and Device Innovation
Orthotic Interventions and Load Distribution
Customized orthoses and exoskeletons have evolved to optimize load distribution across healing bone segments. By offloading stressed regions or modulating joint alignment, these devices support safe progression through weight-bearing milestones. Finely tuned adjustments can reduce deleterious shear forces, thereby enhancing callus formation and consolidation.
3D-Printed Scaffolds and Bioactive Implants
Advances in material science have spawned **bioactive** scaffolds that provide both mechanical support and cellular cues to facilitate bone regeneration. 3D printing enables patient-specific geometries and porosity gradients, promoting vascular ingrowth and osteointegration. Future developments may incorporate growth factors or stem cell seeding to further accelerate healing.
Imaging and Monitoring Technologies
Quantitative imaging tools, such as high-resolution peripheral quantitative computed tomography (HR-pQCT) and dual-energy X-ray absorptiometry (DXA), allow precise tracking of bone mineral density and microarchitecture. Wearable sensors and motion-capture systems provide real-time feedback on mechanical loading during rehabilitation exercises, enabling data-driven adjustments to therapy.
Nutrition and Pharmacologic Support
Dietary Optimization for Bone Healing
Effective rehabilitation extends beyond mechanical interventions; **nutrition** plays a pivotal role. Key dietary components include:
- Protein: Essential for collagen matrix synthesis and muscle repair.
- Calcium and Vitamin D: Fundamental for mineralization and systemic calcium homeostasis.
- Omega-3 Fatty Acids: Anti-inflammatory properties that may modulate bone resorption.
- Micronutrients: Zinc, magnesium, and vitamin K2 support osteoblastic activity.
Pharmacologic Agents in Supportive Care
In certain cases, pharmacologic augmentation may be warranted. Bisphosphonates, selective estrogen receptor modulators, and parathyroid hormone analogues influence remodeling dynamics. While typically reserved for osteoporosis management, off-label applications in rehabilitation settings are under investigation. Careful assessment of risk-benefit profiles is critical, particularly in younger or athletic populations.
Emerging Directions and Research Frontiers
Regenerative Medicine and Cellular Therapies
Stem cell-based approaches aim to deliver osteoprogenitor cells directly to injury sites, offering the potential for enhanced **recovery**. Autologous mesenchymal stem cells, delivered via injection or scaffold implantation, have shown promise in preclinical models. Gene-editing techniques targeting key regulators of bone formation may further refine therapeutic efficacy.
Personalized Rehabilitation Pathways
Integration of patient-specific data—genetic profiles, biomechanical metrics, and lifestyle factors—facilitates personalized rehabilitation algorithms. Machine learning models can predict optimal loading parameters and anticipate complications, enabling proactive adjustments. Telehealth platforms expand access to specialized care, ensuring continuity of therapy beyond the clinical setting.
Translational Challenges and Ethical Considerations
While technological and biological innovations hold great promise, translational hurdles remain. Standardization of outcome measures, long-term safety data, and equitable access to advanced therapies are pressing issues. Ethical frameworks must guide the deployment of novel interventions, balancing innovation with patient autonomy and informed consent.