Physical activity exerts a profound influence on the human skeleton, driving adaptive changes in bone architecture, strength, and resilience. By understanding how mechanical forces interact with the complex hierarchy of bone tissue, researchers and clinicians can develop targeted interventions to prevent osteoporosis, enhance fracture healing, and optimize athletic performance. This article explores the multifaceted relationship between physical activity and bone microstructure, emphasizing the underlying biological mechanisms, variation among exercise modalities, and clinical implications for musculoskeletal health.
Influence of Mechanical Loading on Bone Architecture
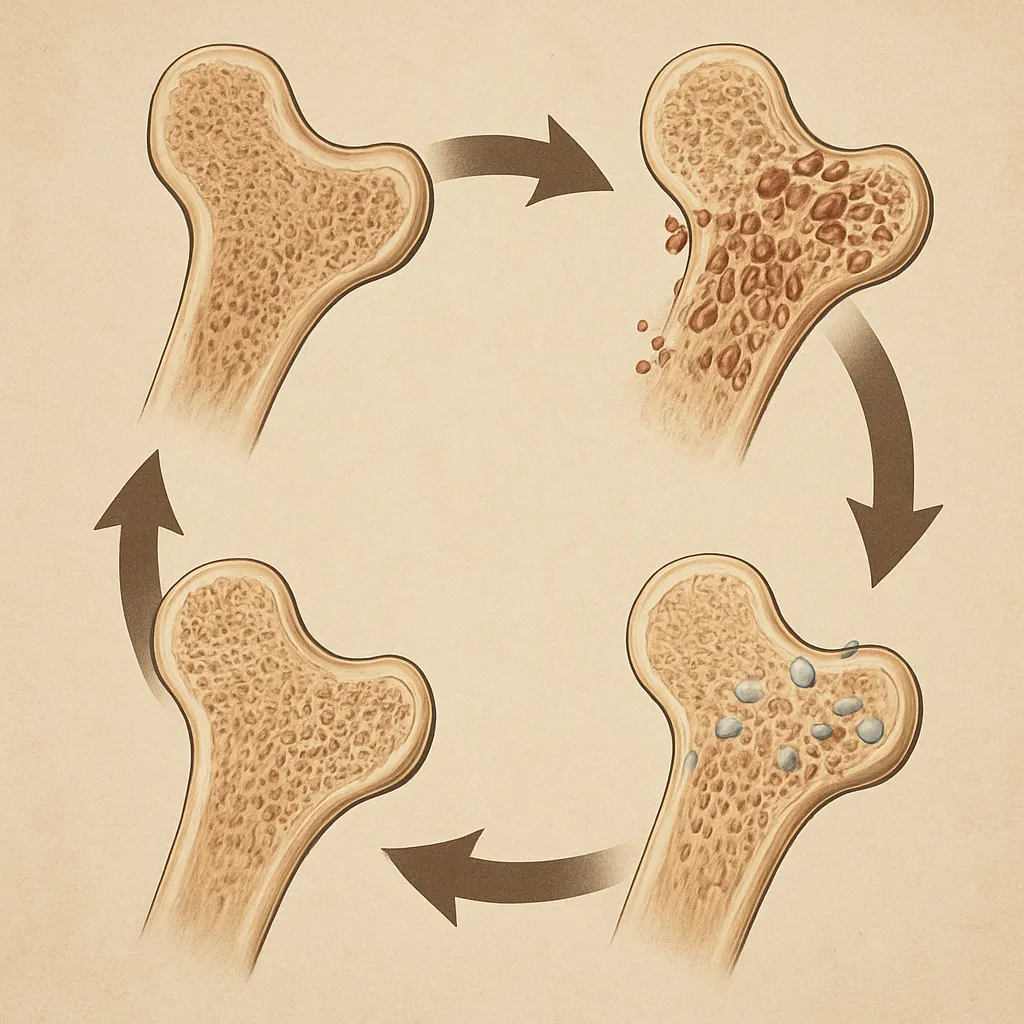
Bones respond dynamically to mechanical forces through a process known as mechanotransduction, wherein physical stimuli are converted into biochemical signals that regulate tissue adaptation. Wolff’s law, first proposed in the 19th century, posits that bone structure adapts to the magnitude and direction of loads applied. Modern imaging techniques, such as high-resolution peripheral quantitative computed tomography (HR-pQCT) and micro-CT, have revealed how repetitive stresses reshape both the cortical and trabecular compartments of bone.
Cortical Adaptations
- Increased periosteal apposition strengthens the outer layer, enhancing resistance to bending and torsion.
- Improved cortical thickness boosts overall density, reducing susceptibility to fractures.
- Microcrack repair mechanisms maintain structural integrity under repeated loading.
Trabecular Remodeling
- Alteration of trabecular orientation aligns struts with predominant force vectors.
- Enhanced connectivity and lower trabecular separation confer greater load distribution capacity.
- Regional variations reflect site-specific mechanical demands, such as in the distal radius versus the femoral neck.
Physical loads induce site-specific bone modeling, where osteocytes act as mechanosensors embedded within the matrix. These cells regulate the balance between bone formation by osteoblasts and bone resorption by osteoclasts, ensuring optimal adaptation to habitual activities.
Cellular Mechanisms Underlying Bone Remodeling
At the cellular level, bone remodeling is orchestrated by a network of signaling pathways that respond to mechanical, hormonal, and nutritional cues. Mechanical strain triggers fluid flow through the lacunar-canalicular system, stimulating mechanosensitivity in osteocytes. Key molecular players include:
- Wnt/β-catenin signaling: Promotes mineralization and osteoblast differentiation.
- RANK/RANKL/OPG axis: Regulates osteoclastogenesis and bone resorption.
- Sclerostin secretion: Acts as a negative regulator, inhibiting bone formation when mechanical stimuli are reduced.
- IGF-1 and growth factors: Enhance cellular proliferation and matrix synthesis in response to exercise.
Integration of these pathways ensures that bone mass and microarchitecture are tailored to the intensity, frequency, and duration of mechanical loads. Emerging evidence suggests that systemic factors, such as inflammatory cytokines and endocrine signals, modulate local remodeling processes, linking overall health status to skeletal adaptation.
Impact of Different Types of Exercise on Bone Microstructure
Not all physical activities yield equivalent benefits to the skeleton. Understanding the specific loading patterns associated with various exercises helps optimize training regimens for bone health.
Weight-Bearing and High-Impact Activities
- Running and jumping sports: Generate rapid ground-reaction forces that stimulate periosteal expansion and trabecular reinforcement.
- Resistance training: Induces localized stress at muscle–bone interfaces, enhancing cortical thickness and increasing mechanical strength.
- Gymnastics and volleyball: Promote site-specific adaptations, particularly at the wrist and spine.
Non-Weight-Bearing and Low-Impact Activities
- Cycling and swimming: Provide cardiovascular benefits but yield minimal osteogenic stimuli in the lower limbs.
- Yoga and Pilates: Improve balance and flexibility, potentially reducing fall risk, yet produce modest changes in density compared to high-impact exercise.
Novel Exercise Interventions
Emerging modalities integrate vibration platforms, whole-body vibration therapy, and functional electrical stimulation to mimic high-frequency mechanical loads. Clinical trials indicate that vibration may enhance trabecular connectivity and cortical mass in populations unable to perform conventional exercise, such as the elderly or immobilized patients.
Clinical Implications and Future Directions
Optimizing bone health through physical activity has far-reaching implications for prevention and treatment of skeletal disorders. Strategies informed by microstructural insights include:
- Personalized exercise prescriptions based on imaging biomarkers of trabecular integrity and cortical geometry.
- Combined pharmacological and mechanical interventions, utilizing anabolic agents in conjunction with tailored loading protocols.
- Early-life exercise programs to maximize peak bone mass and minimize lifelong fracture risk.
- Rehabilitation protocols for fracture healing that balance mechanical stimulation with protection of the injury site.
Advanced computational models, such as finite element analysis, enable simulation of bone adaptation under diverse loading scenarios, guiding both research and clinical decision-making. Integration of wearable sensors and machine learning algorithms promises real-time monitoring of mechanical exposures, facilitating dynamic adjustment of exercise regimens to maximize skeletal benefits.
By elucidating the complex interplay between physical activity and bone microstructure, the medical community moves closer to harnessing nature’s own signals for robust, long-lasting skeletal health. Continued interdisciplinary collaboration will drive innovation in diagnostics, interventions, and public health strategies aimed at preserving bone integrity across the lifespan.