Bone tissue represents a dynamic organ system, constantly adapting to mechanical demands and metabolic signals. The complex interplay between specialized cells, intricate molecular pathways, and mineral deposition events underlies the remarkable strength and resilience of the vertebrate skeleton. This article delves into the biological foundations of bone formation, reveals the stepwise sequence of mineralization, examines the crucial regulatory networks, describes common disorders arising from impaired matrix hardening, and highlights cutting-edge analytical methods that illuminate these processes.
Basic Biology of Bone Tissue
Cellular Components
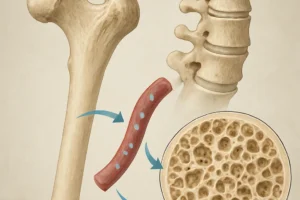
Bone is maintained by a coordinated effort of multiple cell types. Osteoblasts secrete the organic scaffold upon which minerals accumulate. Once embedded in the matrix, some osteoblasts differentiate into osteocytes, forming a sophisticated communication network via dendritic processes. Conversely, osteoclasts are giant, multinucleated cells responsible for resorbing mineralized tissue, balancing ongoing bone growth with controlled degradation.
Extracellular Matrix Composition
The mineralized matrix consists of two primary constituents. The organic fraction is dominated by type I collagen fibrils that provide tensile strength and a template for crystal deposition. Non-collagenous proteins such as osteocalcin, osteonectin, and bone sialoprotein modulate the nucleation sites and crystal morphology. The inorganic fraction is dominated by nanocrystals of hydroxyapatite [Ca10(PO4)6(OH)2], providing compressive rigidity and serving as a reservoir for systemic ions.
Mechanisms of Mineral Deposition
Role of Matrix Vesicles
Mineralization initiates within small, membrane-bound packages known as matrix vesicles. Released by osteoblasts and chondrocytes, these vesicles concentrate calcium and phosphate ions via specific transporters and enzyme-mediated hydrolysis of phosphate donors. The intravesicular environment favors the formation of amorphous calcium phosphate, which matures into crystalline hydroxyapatite before breaching the lipid bilayer and spreading into the extracellular matrix.
Nucleation and Crystal Growth
The transition from amorphous precursors to lattice-structured crystals relies on defined nucleation sites provided by non-collagenous proteins. Osteocalcin and osteopontin bind to collagen surfaces, creating focal points where initial crystallites can anchor and grow. Over time, individual crystals coalesce and align along collagen fibrils, achieving a hierarchical architecture that enhances both toughness and stiffness.
Regulatory Factors and Signaling Pathways
Endocrine Hormones
Bone mineralization is under the influence of systemic hormones that govern calcium-phosphate homeostasis. Parathyroid hormone (PTH) elevates serum calcium by stimulating osteoclast activity and enhancing renal reabsorption. In contrast, calcitonin reduces osteoclast-mediated resorption, favoring net deposition. Active vitamin D (1,25-dihydroxyvitamin D3) promotes intestinal absorption of calcium and phosphate, providing essential building blocks for crystal growth.
Local Growth Factors
Pertinent to skeletal development, bone morphogenetic proteins (BMPs) and transforming growth factor-β (TGF-β) family members orchestrate osteoprogenitor differentiation. These mediators trigger gene expression programs that increase matrix protein synthesis, accelerate vesicle release, and fine-tune mineralization rate. Mechanical loading further modulates local signaling, with fluid flow-induced shear stress potentiating anabolic pathways.
Clinical Implications and Disorders
Defects in any phase of matrix production or mineral nucleation can precipitate skeletal fragility. Common pathologies illustrate how critical each molecular event is:
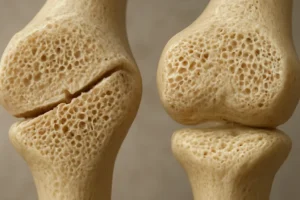
- Osteoporosis – Characterized by diminished bone mass and disrupted microarchitecture, elevating fracture risk in aging populations.
- Osteomalacia and rickets – Results from inadequate mineral availability, often due to vitamin D deficiency or phosphate wasting, leading to softened and deformed bones.
- Osteopetrosis – A genetic disorder marked by impaired osteoclast function, causing overly dense but brittle skeletons prone to cranial nerve compression.
- Hypophosphatasia – Arises from mutations in alkaline phosphatase, hindering matrix vesicle function and preventing proper crystal formation.
Advanced Imaging and Analytical Techniques
To decipher the nanoscale events of bone mineralization, researchers employ a range of sophisticated tools. Transmission electron microscopy (TEM) visualizes initial mineral clusters within vesicles, revealing crystal orientation and size distribution. Fourier transform infrared spectroscopy (FTIR) and Raman microscopy differentiate mineral phases and detect subtle changes in carbonate substitution. Synchrotron-based X-ray diffraction provides high-resolution maps of crystal lattice parameters across tissue sections, linking molecular structure to mechanical performance. Finite element modeling integrates such data, predicting how variations in mineral density and collagen organization translate into macroscopic bone strength.
Integration of Research and Therapeutics
Bridging bench and bedside, insights into the molecular choreography of bone mineralization inform treatment strategies. Bisphosphonates and monoclonal antibodies target resorption pathways, stabilizing skeletal integrity in osteoporosis. Emerging approaches harness BMPs in localized delivery systems to enhance repair in non-union fractures. Ion-substituted biomaterials, enriched with strontium or magnesium, aim to mimic natural bone mineral properties and promote favorable cellular responses when used as implants or scaffolds. As our understanding deepens, personalized interventions that restore the delicate equilibrium of deposition and resorption grow within reach, promising improved outcomes for patients facing skeletal disorders.