Anemia is a common hematological condition that exerts significant effects on the bone marrow microenvironment and its capacity to sustain normal blood cell formation. This article examines how anemia alters erythropoiesis, influences the behavior of hematopoietic stem cells, and disrupts the balance of the bone and medicine interface. By exploring the underlying mechanisms—from iron deficiency and chronic inflammation to hypoxic signaling and cytokine regulation—clinicians and researchers can better appreciate the intricate interplay that shapes marrow function under stressful conditions.
Pathophysiological Mechanisms Linking Anemia to Marrow Dysfunction
The onset of anemia triggers a cascade of adaptive and maladaptive processes within the bone marrow. At its core, anemia is defined by a reduction in red blood cell mass or hemoglobin concentration, leading to tissue hypoxia. In response, marrow niches undergo morphological and molecular changes aimed at restoring oxygen-carrying capacity:
- Erythropoietin Release: Hypoxia-inducible factors (HIFs) stimulate the kidney to produce erythropoietin (EPO), which travels to the marrow and binds receptors on erythroid progenitors.
- Stem Cell Activation: Elevated EPO levels shift the balance of hematopoietic stem cells (HSCs) towards erythroid lineage commitment, potentially at the expense of other lineages, such as myeloid or lymphoid cells.
- Cytokine Network Alterations: Anemic stress often coincides with elevated inflammatory cytokines—interleukin-6 (IL-6), tumor necrosis factor-alpha (TNF-α), and interferon-gamma (IFN-γ)—which can inhibit erythroid maturation and promote myelopoiesis.
These adaptations, while compensatory in the short term, may lead to long-term remodeling of the marrow microenvironment. Progressive changes in stromal cell function, vascular architecture, and extracellular matrix production can compromise the supportive niche required for balanced hematopoiesis.
Iron Deficiency, Inflammation, and Niche Remodeling
One of the most prevalent causes of anemia worldwide is iron deficiency. Iron is essential for hemoglobin synthesis, mitochondrial function, and DNA replication. In iron-deficient states:
- Proerythroblasts accumulate without maturing, as iron availability limits hemoglobinization.
- Macrophages within erythroblastic islands become iron-starved, altering their support of developing erythroid cells.
- Stromal cells reduce secretion of critical growth factors, including stem cell factor (SCF) and vascular endothelial growth factor (VEGF), impairing HSC maintenance and angiogenesis.
Concurrently, chronic inflammation—driven by infections, autoimmune disorders, or malignancies—further perturbs marrow function. Inflammatory cytokines not only sequester iron via upregulation of hepcidin but also induce apoptosis in erythroid progenitors. This phenomenon, often termed “anemia of chronic disease,” exemplifies how immune signals can override homeostatic mechanisms. Prolonged exposure to inflammatory mediators leads to:
- Marrow fibrosis, due to activation of fibroblast-like reticular cells.
- Altered interactions between HSCs and osteoblasts, key regulators of the endosteal niche.
- Impaired angiogenic remodeling, limiting nutrient and oxygen delivery to proliferating progenitors.
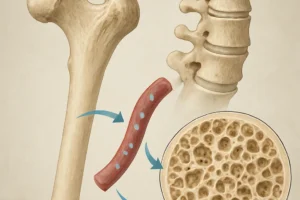
Hypoxia-Induced Signaling and Crosstalk with Bone Cells
The hypoxic conditions inherent to anemia activate a network of transcription factors and paracrine signals that influence both hematopoietic and skeletal components of the marrow. Key aspects include:
- HIF Pathways: HIF-1α and HIF-2α stabilize under low-oxygen conditions, upregulating genes involved in EPO production, iron transport (transferrin receptor), and glycolytic metabolism.
- Osteoblast-HSC Interactions: Osteoblasts, the bone-forming cells on the endosteal surface, express HIF-responsive genes that can modify the quiescence or activation state of adjacent HSCs.
- Angiogenesis: Hypoxia promotes vascular endothelial growth factor (VEGF) secretion, expanding the sinusoidal network that supplies nutrients to maturing blood cells. However, excessive or aberrant angiogenesis can disrupt niche architecture.
Importantly, these hypoxia-driven adaptations are double-edged: while they aim to rectify oxygen deprivation, persistent HIF activation can skew lineage differentiation and foster a microenvironment that supports pathological remodeling, such as myelofibrosis.
Clinical Implications and Therapeutic Strategies
A comprehensive understanding of how anemia influences marrow function informs several therapeutic avenues. Clinicians must consider the multifaceted nature of anemic states when selecting interventions:
Erythropoiesis-Stimulating Agents (ESAs)
By mimicking endogenous EPO, ESAs can accelerate red blood cell production. Yet their use must be balanced against risks such as hypertension, thromboembolism, and potential exacerbation of underlying malignancies. Close monitoring of hemoglobin levels and iron status is essential during therapy.
Iron Supplementation and Modulation of Hepcidin
Oral or intravenous iron can rectify deficiency, but in inflammatory anemia, elevated hepcidin impedes iron absorption and release from macrophages. Novel hepcidin antagonists and ferroportin agonists are under investigation to overcome this barrier.
Anti-Inflammatory and Immunomodulatory Approaches
Targeting cytokines such as IL-6 (with monoclonal antibodies) or TNF-α inhibitors can alleviate anemia of chronic disease by reducing inflammatory suppression of erythropoiesis. Additionally, small-molecule modulators of the JAK-STAT pathway show promise in treating myelofibrosis and related conditions.
Bone-Targeted Therapies
Given the crosstalk between bone cells and hematopoiesis, agents that enhance osteoblast function—such as parathyroid hormone analogs—or inhibit osteoclast-mediated resorption may help restore niche integrity. Trials are exploring the use of sclerostin inhibitors to promote healthy bone remodeling and support HSC maintenance.
Future Directions and Research Frontiers
Ongoing research aims to unravel the complex network of signals that govern marrow adaptation to anemia. Emerging topics include:
- Single-cell transcriptomic mapping of HSCs and niche cells under anemic stress.
- Gene-editing strategies to correct inherited red cell disorders and enhance resilience to hypoxia.
- Bioengineered scaffolds that mimic the marrow microenvironment for ex vivo expansion of HSCs.
- Integration of metabolic profiling to identify novel regulators of erythropoiesis and niche health.
By bridging advances in molecular biology, bioengineering, and clinical medicine, the field moves closer to tailored therapies that not only address the symptoms of anemia but also restore the dynamic equilibrium of the bone marrow ecosystem.