Bone remodeling cycles represent a continuous, highly regulated process through which the skeletal system maintains its integrity, adapts to mechanical demands, and repairs microdamage. This intricate interplay of cellular activity ensures that old or damaged bone is replaced by new tissue, preserving structural strength and calcium homeostasis. Understanding the critical phases of this process reveals insights into conditions such as osteoporosis, fracture healing, and metabolic bone diseases.
Cellular Actors and Molecular Signals
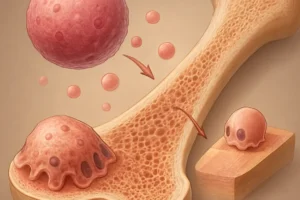
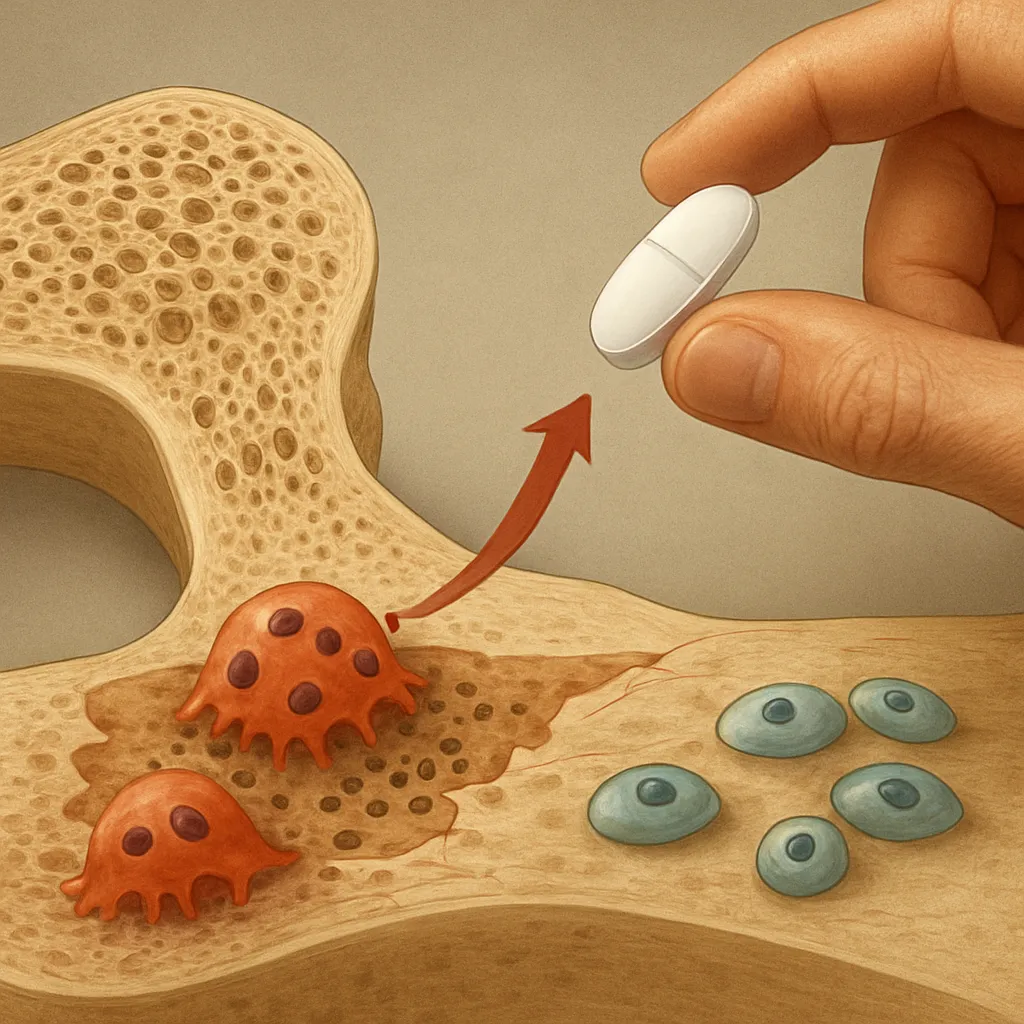
The dynamic balance between bone formation and resorption relies primarily on two specialized cell types: osteoclasts and osteoblasts. Osteoclasts are multinucleated cells responsible for bone resorption, degrading the mineralized matrix, while osteoblasts lay down new bone tissue. A third cell type, the osteocyte, serves as a mechanosensor and orchestrates signaling pathways that couple resorption to formation.
- Osteoclast Differentiation: Derived from hematopoietic precursors, osteoclast maturation is driven by receptor activator of nuclear factor kappa-B ligand (RANKL) and macrophage colony-stimulating factor (M-CSF). Binding of RANKL to its receptor RANK triggers the NF-κB pathway, promoting osteoclast fusion and activation.
- Osteoblast Function: Originating from mesenchymal stem cells (MSCs), osteoblasts secrete collagen type I and non-collagenous proteins, initiating matrix mineralization. Key transcription factors like Runx2 and osterix regulate osteoblast differentiation.
- Osteocyte Signaling: Embedded within the bone matrix, osteocytes respond to mechanical strain by releasing sclerostin and nitric oxide, which modulate osteoblast activity and maintain bone homeostasis.
Communication among these cells involves a network of cytokines, growth factors, and hormones. Parathyroid hormone (PTH) and vitamin D enhance RANKL expression, tipping the scale toward resorption when calcium levels are low. Conversely, calcitonin inhibits osteoclast activity, reducing bone breakdown.
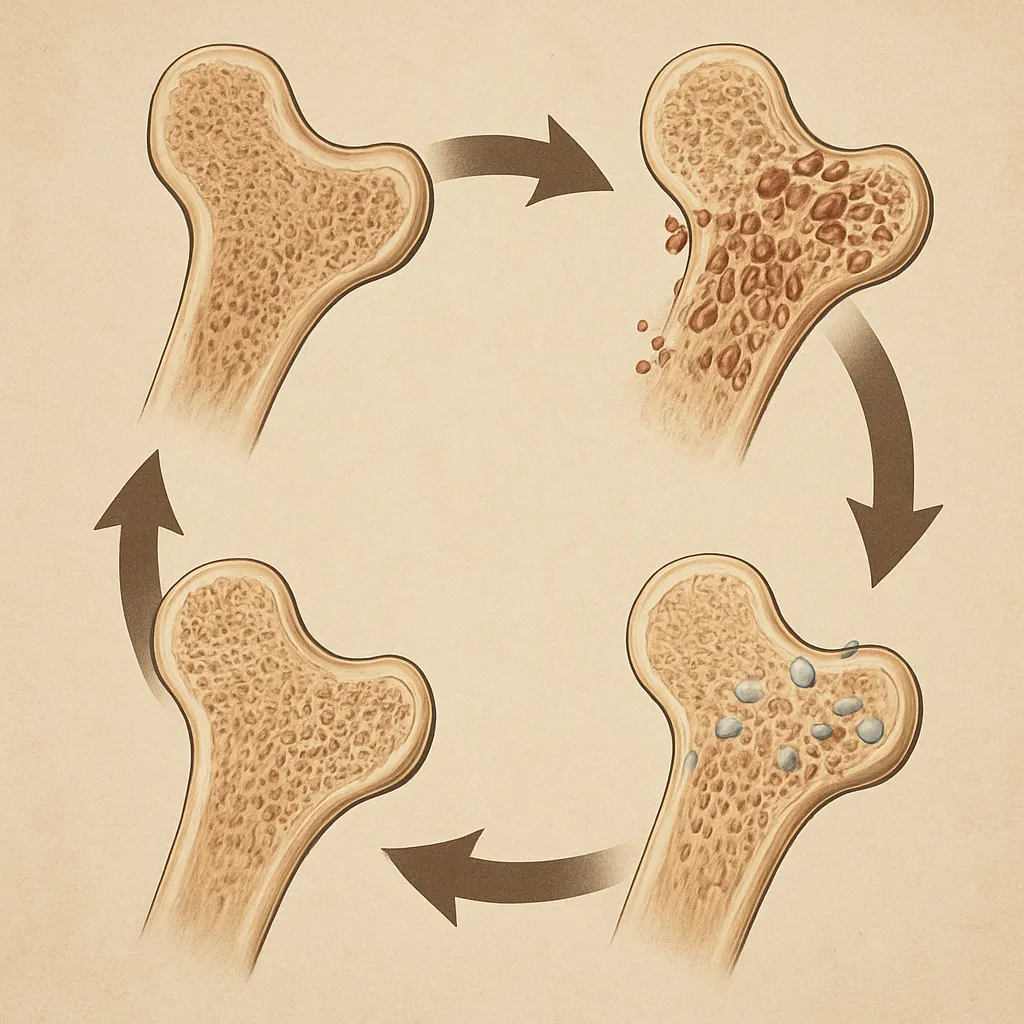
Phases of the Remodeling Cycle
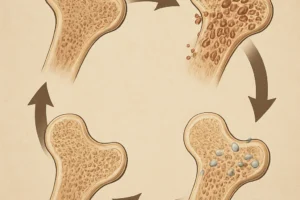
A complete remodeling cycle encompasses four distinct but overlapping stages: activation, resorption, reversal, and formation. Each phase is orchestrated by precise molecular signaling to ensure seamless transition from bone removal to renewal.
Activation
Mechanical microdamage or systemic signals initiate activation. Osteocytes detect microcracks and secrete factors such as RANKL and monocyte chemoattractant protein-1 (MCP-1), recruiting osteoclast precursors to the site. Local release of prostaglandins and interleukin-6 (IL-6) amplifies this recruitment.
Resorption
Once differentiated, osteoclasts attach to the bone surface, creating a sealed resorption lacuna. They secrete hydrogen ions and proteolytic enzymes like cathepsin K to dissolve the inorganic and organic components of the bone matrix. This process liberates calcium and phosphate into the bloodstream.
- Acidification of the lacuna by the vacuolar H+-ATPase pump.
- Enzymatic degradation of collagen fibers.
- Release of growth factors embedded in the matrix, such as transforming growth factor beta (TGF-β).
Reversal
Following resorption, the reversal phase resets the surface for new bone deposition. Mononuclear cells remove residual debris, while local factors like insulin-like growth factor-1 (IGF-1) and bone morphogenetic proteins (BMPs) attract osteoblast progenitors.
Formation
Osteoblasts synthesize osteoid, a collagen-rich, unmineralized matrix. Over days to weeks, mineral crystals precipitate, stiffening the tissue. A subset of osteoblasts becomes embedded, differentiating into osteocytes, while others undergo apoptosis or revert to bone lining cells.
- Synthesis of type I collagen and osteocalcin.
- Matrix vesicle release and nucleation of hydroxyapatite.
- Regulation by local growth factors and mechanical loading.
Regulation of Bone Remodeling
The remodeling process is fine-tuned by systemic and local regulators that ensure bone adapts to physiological demands. Disruption in these controls leads to pathological conditions characterized by loss of bone mass or excessive build-up.
- Endocrine Control: PTH, secreted in response to hypocalcemia, promotes resorption, whereas estrogen preserves bone mass by inhibiting osteoclastogenesis. Decline in estrogen during menopause accelerates bone loss.
- Mechanical Loading: Wolff’s law describes how bone adapts its architecture to loading patterns. Compressive forces stimulate osteocytes to downregulate sclerostin, thereby enhancing formation.
- Nutritional Factors: Adequate intake of calcium, phosphate, vitamin D, and protein is essential. Deficiencies impair mineralization, leading to osteomalacia or rickets.
- Inflammatory Mediators: Chronic inflammation elevates cytokines like tumor necrosis factor-alpha (TNF-α), favoring resorption and contributing to bone erosions in rheumatoid arthritis.
Clinical Implications and Therapeutic Targets
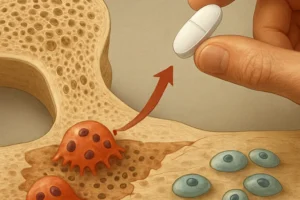
A clear grasp of bone remodeling underpins strategies for treating metabolic bone disorders, fracture repair, and implant integration. Modulating the resorption-formation balance through targeted therapies has revolutionized patient outcomes.
- Antiresorptive Agents: Bisphosphonates bind to bone mineral and inhibit osteoclast-mediated resorption. Denosumab, a monoclonal antibody against RANKL, reduces osteoclast activity and prevents fractures.
- Anabolic Treatments: PTH analogs, such as teriparatide, intermittently stimulate osteoblasts more than osteoclasts, promoting net bone gain. Sclerostin inhibitors (e.g., romosozumab) enhance formation by preventing Wnt pathway antagonism.
- Biomaterials and Scaffolds: In bone tissue engineering, biocompatible scaffolds deliver cells and growth factors to critical-sized defects, facilitating new bone growth and vascularization.
- Diagnostic Markers: Biochemical markers like serum C-terminal telopeptide (CTX) and procollagen type I N-terminal propeptide (P1NP) reflect resorption and formation rates, respectively, guiding treatment efficacy.
Emerging research explores the role of microRNAs and epigenetic regulation in bone remodeling, unlocking potential for precision medicine. Understanding how these pathways integrate with mechanical and hormonal cues will pave the way for novel interventions.
Challenges and Future Directions
Despite significant advances, achieving a complete restoration of bone architecture remains challenging. Factors such as aging, diabetes, and sustained inflammation impair remodeling efficiency, resulting in compromised healing and increased fracture risk.
- Identifying patient-specific remodeling profiles through advanced imaging and molecular diagnostics.
- Developing combination therapies that couple anabolic and antiresorptive mechanisms.
- Harnessing stem cell approaches to replenish osteoprogenitor pools in degenerative disorders.
- Optimizing biomaterial scaffolds with controlled release of signaling molecules to mimic natural microenvironments.
As the global burden of osteoporosis and skeletal injuries rises, interdisciplinary collaboration among biologists, engineers, and clinicians will be critical. By elucidating the complex network of signals driving bone remodeling, the next generation of therapies aims to restore skeletal health and improve quality of life.