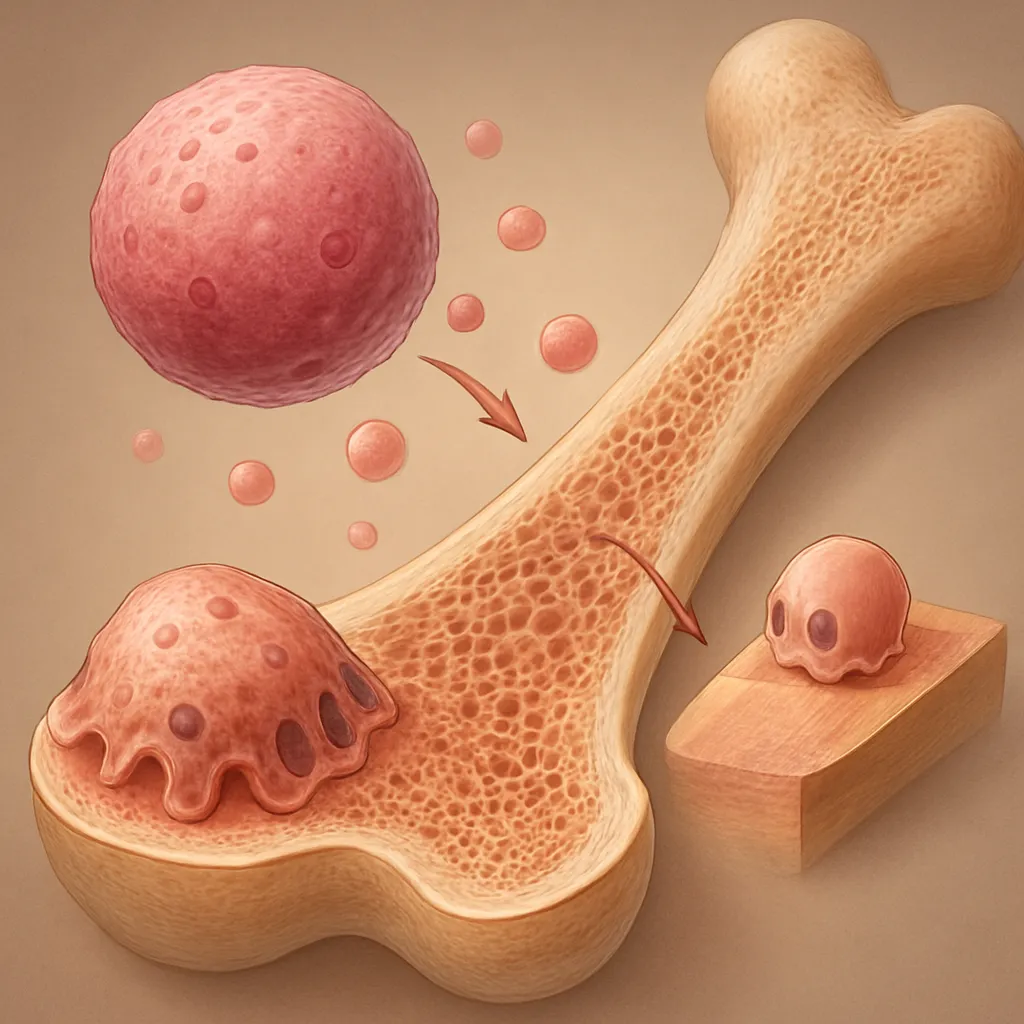
The intricate balance between bone formation and resorption is orchestrated by a network of chemical messengers that regulate cellular activity and tissue homeostasis. Among these, cytokines play a pivotal role in directing the behavior of bone cells, modulating immune responses, and influencing the overall skeletal health. Understanding the contributions of these signaling molecules has profound implications for treating metabolic bone disorders and optimizing regenerative therapies.
Cytokine Families and Their Functions in Bone Remodeling
Bone remodeling represents a continuous, dynamic process where old bone is removed and replaced with new bone matrix. Two primary cell types coordinate this cycle: osteoclasts, responsible for bone resorption, and osteoblasts, which drive bone formation. A sophisticated interplay of cytokines governs their activity, ensuring skeletal integrity over a lifetime.
Pro-Resorptive Mediators
- RANKL (Receptor Activator of Nuclear Factor κB Ligand): A key promoter of osteoclast differentiation and activation. Binding to its receptor RANK on osteoclast precursors triggers a cascade of signaling events culminating in bone resorption.
- TNF-alpha (Tumor Necrosis Factor-alpha): Potently enhances osteoclastogenesis, often in synergy with RANKL. Elevated levels contribute to pathological bone loss in rheumatoid arthritis and osteoporosis.
- IL-1 (Interleukin-1): Augments both RANKL expression in stromal cells and osteoclast precursor sensitivity. It also stimulates the release of matrix metalloproteinases, facilitating matrix degradation.
Pro-Formative Mediators
- OPG (Osteoprotegerin): Functions as a decoy receptor for RANKL, sequestering it away from RANK and thereby inhibiting osteoclastogenesis. The OPG/RANKL ratio is critical for maintaining bone mass.
- IL-6 (Interleukin-6): Exhibits dual roles. Under physiological conditions, it supports osteoblast differentiation and mineralization. However, chronic elevation can shift the balance toward resorption.
- IL-10: An anti-inflammatory cytokine that suppresses osteoclast formation and activity. Promotes a regenerative environment conducive to bone deposition.
Molecular Mechanisms of Cytokine-Mediated Bone Regulation
The mechanistic pathways through which cytokines influence bone cells involve a series of receptor-ligand interactions, intracellular kinases, transcription factors, and gene expression programs. Deciphering these networks reveals potential targets for therapeutic intervention.
RANK/RANKL/OPG Axis
At the core of osteoclast differentiation lies the RANK/RANKL/OPG system. RANKL produced by osteoblasts or stromal cells engages RANK on monocyte/macrophage precursors, initiating recruitment of adaptor proteins such as TRAF6. Subsequent activation of NF-κB and MAPK pathways induces transcription factors like NFATc1, master regulator of osteoclastogenesis. Homeostasis is preserved when OPG binds RANKL, preventing its interaction with RANK.
MAPK and NF-κB Pathways
Signaling cascades triggered by cytokine receptors converge on MAPK (ERK, JNK, p38) and NF-κB modules. For instance, TNF-alpha binding to TNFR1 activates the IKK complex, leading to IκB degradation and nuclear translocation of NF-κB subunits. This promotes transcription of genes essential for osteoclast survival and function.
JAK/STAT Pathway
Several interleukins signal via the JAK/STAT axis. IL-6 associates with its receptor complex, inducing JAK-mediated STAT3 phosphorylation. STAT3 dimers translocate to the nucleus, upregulating genes implicated in osteoblast proliferation and cytokine production. Dysregulation of JAK/STAT contributes to aberrant bone remodeling in inflammatory states.
Wnt/β-Catenin Signaling
Though primarily known for its role in embryonic development, Wnt signaling is also modulated by cytokines. Pro-inflammatory mediators like TNF-alpha can increase expression of Wnt antagonists (e.g., DKK1, sclerostin), hindering osteoblast maturation. Conversely, anti-inflammatory cytokines may enhance Wnt ligand expression, promoting bone formation.
Clinical Implications and Therapeutic Strategies
Insights into cytokine biology have revolutionized approaches to treating bone disorders. By targeting specific mediators or their receptors, modern therapies aim to restore the equilibrium between resorption and formation.
Biological Inhibitors
- Denosumab: A monoclonal antibody against RANKL, effectively reducing osteoclast activity and fracture risk in osteoporosis patients.
- Anti-TNF Agents: Infliximab, etanercept, and adalimumab neutralize TNF-alpha, alleviating bone loss in rheumatoid arthritis and ankylosing spondylitis.
- IL-6 Receptor Blockers: Tocilizumab inhibits IL-6 signaling, mitigating both joint destruction and systemic bone resorption.
Small Molecule Modulators
- JAK Inhibitors: Tofacitinib and baricitinib interfere with JAK/STAT pathways, reducing inflammatory cytokine production and preserving bone density.
- Sclerostin Antibodies: Romosozumab enhances Wnt signaling by neutralizing sclerostin, resulting in increased bone formation and strength.
Emerging Cell-Based Therapies
Mesenchymal stem cells (MSCs) secrete a myriad of cytokines with immunomodulatory and anabolic effects. MSC transplantation has shown promise in preclinical models of osteoporosis and bone defects by providing a sustained source of regenerative factors.
Nutrition and Lifestyle Modulation
Dietary components can influence cytokine networks. Omega-3 fatty acids and vitamin D exert anti-inflammatory actions, lowering circulating TNF-alpha and IL-6. Regular weight-bearing exercise also modulates cytokine profiles, enhancing osteoblast activity and attenuating resorption.
Future Directions in Cytokine Research
ongoing research aims to elucidate novel cytokines and their interactions within the bone microenvironment. High-throughput omics technologies and advanced imaging are uncovering previously unrecognized signaling nodes. Personalized medicine approaches will tailor anti-cytokine therapies based on patient-specific profiles, optimizing efficacy and minimizing adverse effects. The integration of biomaterials that locally release cytokine modulators offers an exciting frontier for targeted bone regeneration.
Collectively, the dynamic interplay of cytokines constitutes a fundamental regulatory system for skeletal health. Continued exploration of these molecular dialogues holds the key to innovative solutions for debilitating bone diseases.