The skeletal system serves far more than a mere framework for the body; it functions as a dynamic reservoir of essential minerals and actively contributes to systemic physiology. Through intricate interactions between bone cells, extracellular matrix, and the circulatory system, the skeleton maintains mineral equilibrium, supports hematopoiesis, and responds to metabolic demands. This article explores the multifaceted contributions of bone to mineral homeostasis, examining cellular mechanisms, hormonal regulation, and clinical implications in the context of bone and medicine.
Bone as a Mineral Reservoir
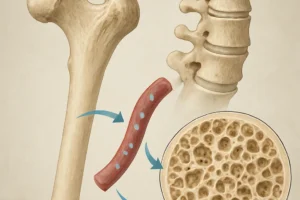
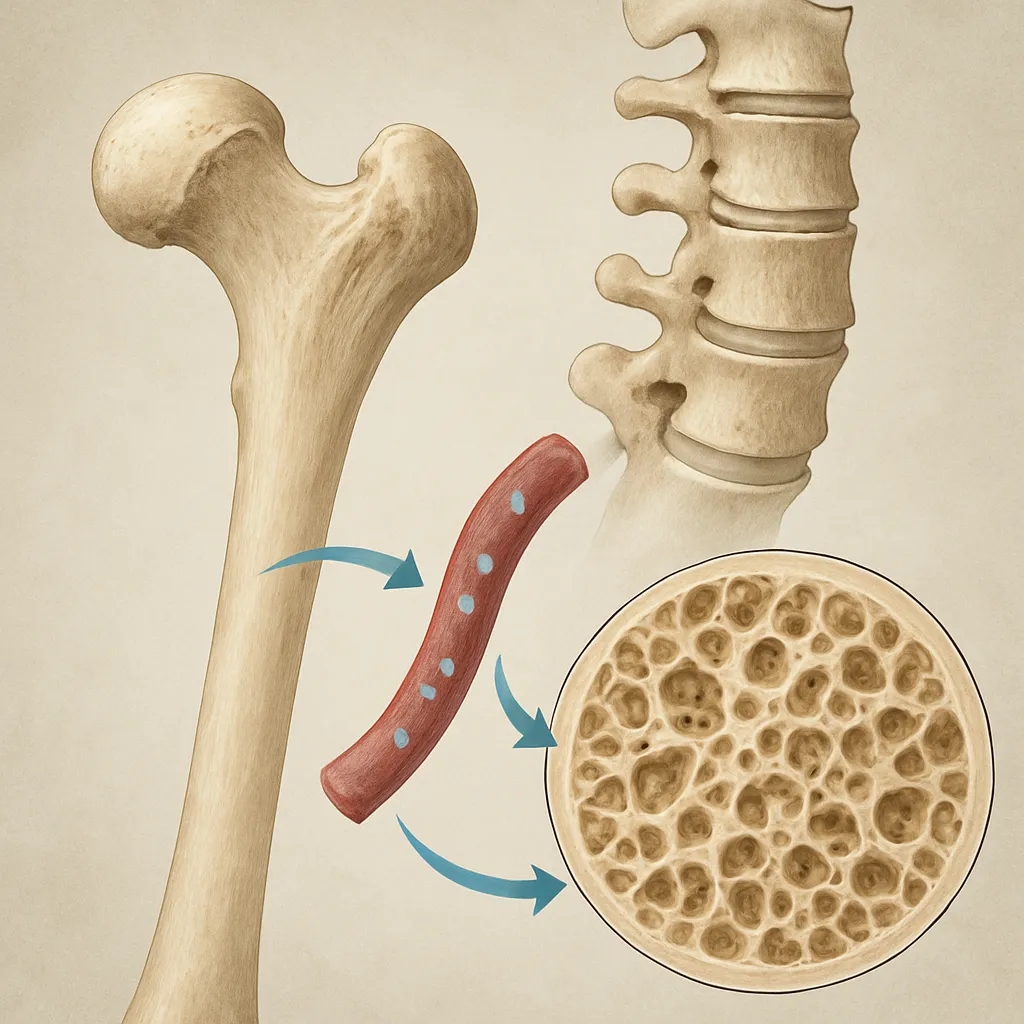
Bone tissue is composed of an organic matrix—predominantly collagen—and a mineral phase rich in hydroxyapatite crystals. The dualistic nature of bone confers tensile strength while accommodating a vast store of calcium and phosphate. Approximately 99% of total body calcium resides within the skeletal compartment, highlighting bone’s pivotal role in maintaining serum mineral levels.
- Composition: Collagen type I fibers embedded in a mineral lattice.
- Storage capacity: Dynamic exchange of ions between bone surfaces and extracellular fluid.
- Mechanical support: Structural integrity enables locomotion and load bearing.
Bone surfaces act as sites of continuous ion exchange, whereby minerals can be released into or sequestered from the bloodstream to uphold homeostasis. The balance between deposition and resorption ensures that mineral demands—whether for neuronal transmission, muscle contraction, or blood coagulation—are reliably met.
Cellular Mechanisms of Mineral Regulation
At the heart of bone’s regulatory capacity lie three principal cell types: osteoblasts, osteoclasts, and osteocytes. Their coordinated activities govern the processes of bone formation, resorption, and the subtle remodeling required to adapt to physiological stresses.
Osteoblasts: Architects of Mineralization
Derived from mesenchymal stem cells, osteoblasts synthesize the organic matrix and initiate mineral deposition. These cells concentrate calcium and phosphate locally, facilitating crystal nucleation and the growth of hydroxyapatite. As mineralization advances, osteoblasts may become embedded in lacunae, differentiating into osteocytes.
Osteoclasts: Specialized Resorbers
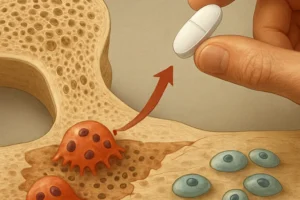
Multinucleated and derived from hematopoietic precursors, osteoclasts attach to bone surfaces, creating an acidic microenvironment that dissolves the mineral phase. By secreting protons and proteolytic enzymes like cathepsin K, they liberate calcium and phosphate into the circulation, modulating blood levels in response to systemic cues.
Osteocytes: Master Regulators
Once thought to be passive, osteocytes form an extensive lacuno-canalicular network that senses mechanical load and chemical changes. They secrete signaling molecules—such as sclerostin—that influence both osteoblast and osteoclast activity, thus fine-tuning the balance of remodeling and ensuring bone adaptation to mechanical demands.
Hormonal Control of Mineral Homeostasis
The endocrine system orchestrates bone’s mineral exchange through several hormones, most notably parathyroid hormone (PTH), vitamin D, and calcitonin. These factors act in concert to preserve optimal serum concentrations of calcium and phosphate.
- Parathyroid Hormone (PTH): Secreted by the parathyroid glands in response to low plasma calcium, PTH enhances osteoclastic resorption indirectly via osteoblast-derived RANKL, boosting calcium release. It also promotes renal calcium reabsorption and stimulates the conversion of vitamin D to its active form.
- Vitamin D (Calcitriol): The active form of vitamin D increases intestinal absorption of calcium and phosphate, while at moderate concentrations it may synergize with PTH to mobilize bone mineral. Its role is critical in maximizing dietary mineral uptake to support both skeletal and extraskeletal demands.
- Calcitonin: Secreted by thyroid parafollicular cells when calcium levels rise, calcitonin directly inhibits osteoclast activity, reducing bone resorption and facilitating mineral deposition within bone tissue.
These hormonal pathways underscore the skeleton’s integration into a broader endocrine network. Disruption of any component—whether due to glandular dysfunction, nutritional deficiencies, or genetic variants—can precipitate imbalances in bone mineral density and systemic mineral equilibrium.
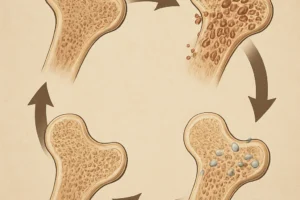
Bone Microarchitecture and Remodeling Sites
Bone remodeling occurs at discrete sites called basic multicellular units (BMUs). Within each BMU, a cohort of osteoclasts excavates a resorption pit, followed by osteoblast-mediated refilling. The orchestration of this process ensures uniform renewal of both cortical and trabecular compartments, preserving microarchitecture and mechanical competence.
- Cortical bone: Dense outer shell that undergoes slow remodeling, critical for fracture resistance.
- Trabecular bone: Highly vascular, exhibits rapid turnover to meet metabolic needs and adapt to mechanical load.
Clinical Perspectives in Bone Mineral Homeostasis
Understanding the skeletal contributions to mineral balance has direct medical relevance. Disorders such as osteoporosis, rickets, and chronic kidney disease-mineral and bone disorder (CKD-MBD) exemplify how impaired regulation can lead to structural weakness, deformity, and systemic complications.
Osteoporosis and Fracture Risk
Characterized by decreased bone mass and altered microarchitecture, osteoporosis elevates fracture susceptibility. Therapeutic strategies target both bone formation—via agents that stimulate osteoblast function—and resorption—through inhibitors of osteoclast-mediated mineral release.
Renal Osteodystrophy
In chronic kidney disease, disturbed phosphate excretion and dysregulated vitamin D activation lead to secondary hyperparathyroidism and aberrant bone turnover. Management requires careful modulation of dietary phosphate, PTH analogs, and vitamin D supplementation to restore mineral homeostasis.
Emerging Therapies
- Monoclonal antibodies against sclerostin to enhance bone formation.
- Selective PTH receptor modulators to achieve anabolic effects with reduced resorptive activity.
- Nanotechnology approaches for targeted drug delivery within bone microenvironments.
Advances in molecular biology and imaging continue to refine our understanding of the skeletal system’s role in mineral regulation, paving the way for innovative interventions that preserve bone health and systemic equilibrium.