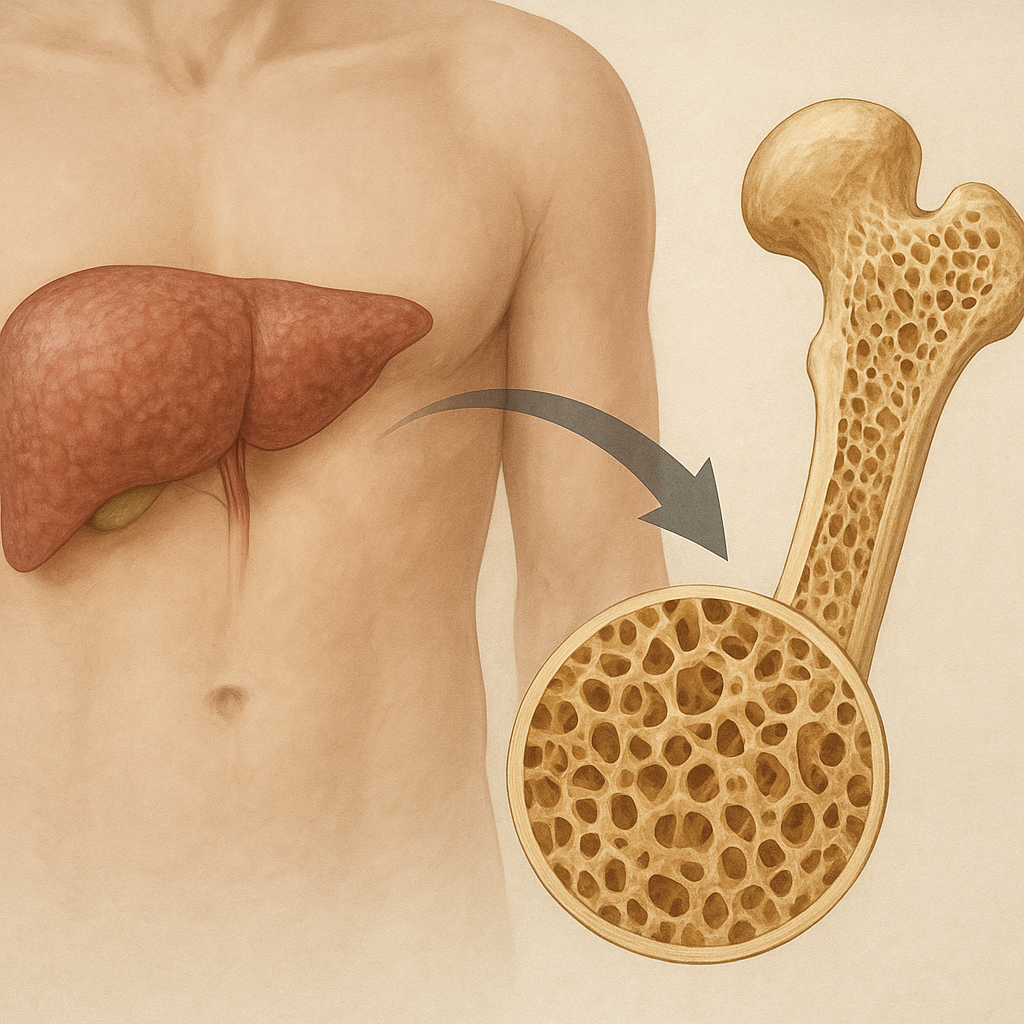
Chronic liver disease exerts a profound impact on bone metabolism, leading to a spectrum of skeletal alterations collectively known as hepatic osteodystrophy. The liver’s pivotal role in nutrient processing, hormone synthesis, and detoxification means that persistent hepatic dysfunction can disrupt the delicate balance between bone formation and resorption. Patients with advanced liver pathology frequently present with reduced bone mineral density, increased fracture risk, and histological changes that resemble both osteoporosis and osteomalacia. Understanding the multifactorial mechanisms behind this interplay is crucial for clinicians and researchers seeking to mitigate the skeletal complications of liver disease.
Pathophysiological Mechanisms
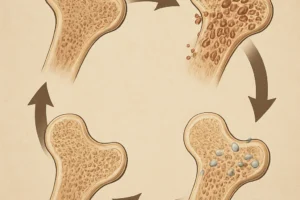
The skeletal system relies on a dynamic equilibrium between the activities of osteoblasts (bone-forming cells) and osteoclasts (bone-resorbing cells). Chronic liver disease disrupts this equilibrium through multiple interconnected pathways.
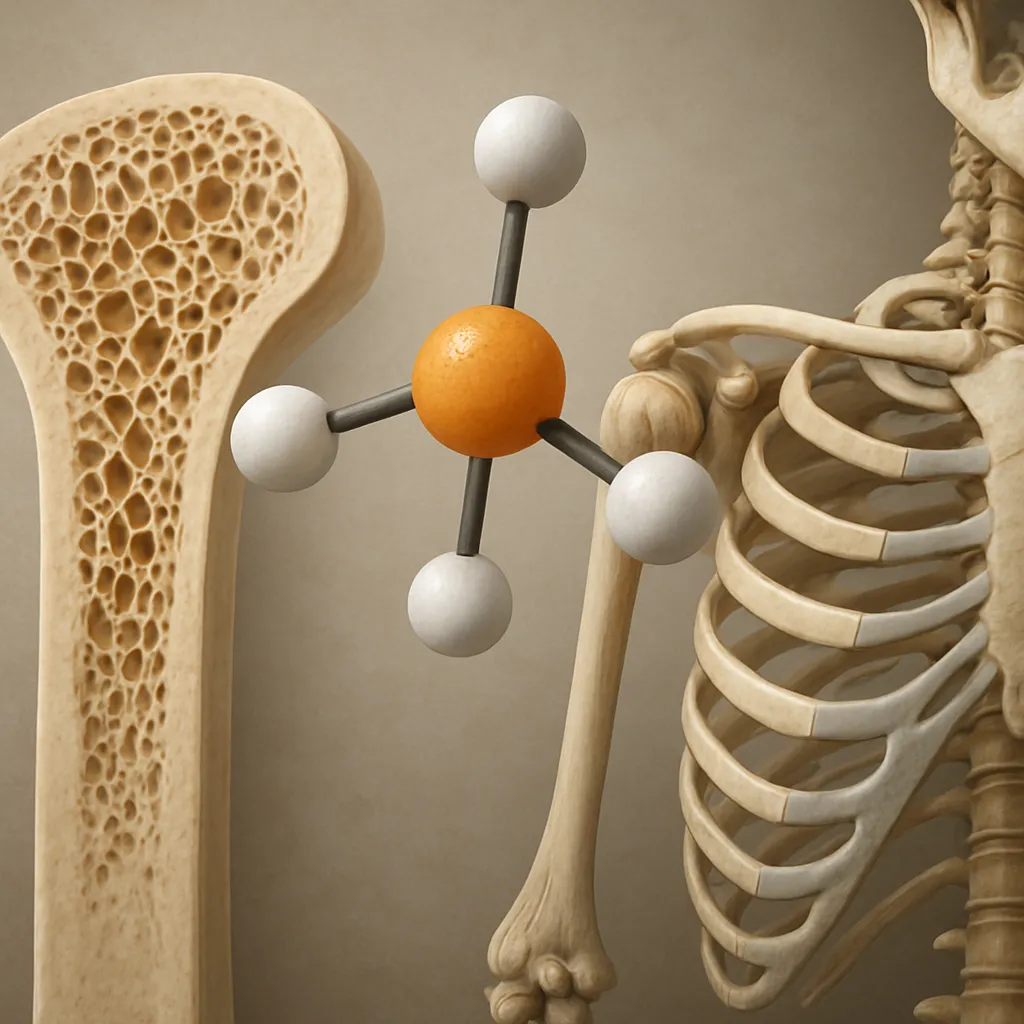
Altered Vitamin D Metabolism
- The liver converts cholecalciferol into 25-hydroxyvitamin D, the main circulating form. Impaired hepatic function leads to inadequate 25-hydroxylation.
- Low levels of 25-hydroxyvitamin D reduce intestinal absorption of calcium and phosphorus, precipitating secondary hyperparathyroidism.
- Deficient vitamin D stores compromise mineralization of the osteoid matrix, promoting osteomalacia-like changes.
Inflammatory Cytokine Overproduction
- Chronic liver injury triggers systemic release of cytokines such as tumor necrosis factor-alpha (TNF-α) and interleukin-6 (IL-6).
- These proinflammatory mediators upregulate osteoclastogenesis via RANK/RANKL signaling, accelerating bone resorption.
- Persistent inflammation impairs osteoblast differentiation and function, further tilting the balance toward net bone loss.
Hormonal Dysregulation
- Reduced hepatic synthesis of insulin-like growth factor-1 (IGF-1) compromises anabolic signaling in bone tissue.
- Altered sex hormone metabolism, including decreased estrogen and testosterone levels, diminishes protective effects on bone density.
- Elevated circulating parathyroid hormone (PTH) due to hypocalcemia exacerbates bone resorption.
Nutritional Deficiencies and Malabsorption
- Malnutrition in advanced liver disease often leads to protein-calorie deficits, essential for collagen matrix formation.
- Fat malabsorption in cholestatic conditions impairs uptake of fat-soluble vitamins (A, D, E, K), crucial for skeletal health.
- Micronutrient shortages, including magnesium and zinc, further compromise bone remodeling processes.
Clinical Manifestations and Diagnostic Approaches
The skeletal consequences of chronic liver disease may remain subclinical until advanced bone loss triggers symptoms. Early recognition facilitates timely intervention.
Presentation and Risk Factors
- Patients often report diffuse bone pain, height loss, or kyphotic posture due to vertebral compression fractures.
- Fracture incidence is elevated at hip, wrist, and spine, correlating with the severity and duration of liver dysfunction.
- Risk factors include cholestatic disorders, cirrhosis, prolonged corticosteroid therapy, and nutritional insufficiencies.
Imaging and Bone Mineral Density Assessment
- Dual-energy X-ray absorptiometry (DEXA) remains the gold standard for quantifying bone mineral density (BMD).
- Quantitative computed tomography (QCT) provides volumetric BMD data and insights into trabecular versus cortical bone compartments.
- Ultrasound-based techniques at peripheral sites may serve as screening tools in resource-limited settings.
Biochemical Markers
- Serum levels of 25-hydroxyvitamin D, PTH, alkaline phosphatase (ALP), and IGF-1 offer clues to underlying metabolic disturbances.
- Bone turnover markers, such as serum C-terminal telopeptide (CTX) and procollagen type I N-terminal propeptide (P1NP), reflect dynamic changes in resorption and formation.
- Markers of cholestasis (bilirubin, gamma-glutamyl transferase) and synthetic function (albumin, prothrombin time) gauge hepatic status contributing to skeletal risk.
Management Strategies
Effective care demands a multidisciplinary approach addressing both liver pathology and bone fragility. Therapeutic decisions hinge on individual patient risk profiles and the stage of liver disease.
Optimizing Nutritional Status
- Ensure adequate protein intake (1.2–1.5 g/kg/day) to support collagen synthesis and muscle mass maintenance.
- Supplement fat-soluble vitamins, particularly vitamin D (800–2,000 IU daily) and calcium (1,000–1,500 mg daily), to correct deficiencies.
- Address malabsorption with medium-chain triglycerides or pancreatic enzyme replacement in cholestatic patients.
Pharmacological Interventions
- Bisphosphonates (alendronate, risedronate) inhibit osteoclast-mediated resorption and have shown efficacy in improving BMD in liver disease cohorts.
- Denosumab, a monoclonal antibody against RANKL, offers an alternative when bisphosphonates are contraindicated.
- Teriparatide, a recombinant PTH fragment, promotes bone formation and may benefit select patients with severe osteoporosis.
Exercise and Rehabilitation
- Weight-bearing activities (resistance training, brisk walking) stimulate osteoblastic activity and enhance bone strength.
- Balance and proprioception exercises reduce fall risk and subsequent fractures.
- Physical therapy tailored to hepatic patients accounts for fatigue and muscle weakness common in chronic liver dysfunction.
Addressing Underlying Liver Disease
- Treat viral hepatitis, autoimmune hepatitis, and cholestatic disorders to slow progression of hepatic injury.
- Evaluate candidacy for liver transplantation, which may partially reverse bone loss over time.
- Minimize exposure to hepatotoxic medications while maintaining fracture prevention regimens.
Monitoring and Long-Term Follow-Up
- Repeat BMD measurements every 1–2 years to assess therapeutic response and adjust treatment.
- Regularly review biochemical markers to ensure nutritional repletion and monitor disease activity.
- Coordinate care among hepatologists, endocrinologists, nutritionists, and physical therapists for comprehensive management.