Bone health depends on a precise balance between formation and resorption, processes collectively known as bone turnover. Understanding this dynamic interplay involves measuring specific biochemical markers that reflect the activity of bone‐forming and bone‐resorbing cells. These indicators provide clinicians with valuable insights into skeletal integrity, disease progression, and treatment efficacy.
Physiology of Bone Remodeling
Bone Cells and Their Roles
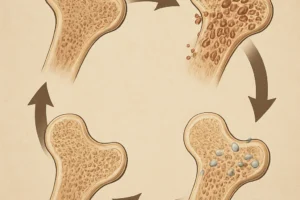
The skeleton continually undergoes renewal through the coordinated actions of osteoclasts and osteoblasts. Osteoclasts are specialized cells responsible for resorption, breaking down mineralized matrix and releasing calcium and phosphate into circulation. In contrast, osteoblasts synthesize new bone matrix, depositing collagen and facilitating mineralization. This coupled activity ensures that microdamage is repaired and mechanical strength is maintained.
Homeostatic Regulation
Maintenance of skeletal integrity demands tight regulatory control. Systemic hormones such as parathyroid hormone (PTH), calcitonin, and vitamin D metabolites modulate cell differentiation and activity. Locally, the RANK/RANKL/OPG axis plays a pivotal role: RANKL (Receptor Activator of Nuclear Factor κB Ligand) expressed by osteoblasts binds RANK on osteoclast precursors, promoting their maturation. Osteoprotegerin (OPG) acts as a decoy receptor, sequestering RANKL and inhibiting osteoclastogenesis. Additional cytokines, growth factors, and mechanical stimuli integrate to fine‐tune this process, preserving skeletal homeostasis.
Key Bone Turnover Markers
Resorption Markers
Markers of bone resorption reflect osteoclast activity and organic matrix degradation. Commonly measured analytes include:
- CTX (C-terminal telopeptide of type I collagen): Released into circulation when osteoclasts degrade bone collagen. Widely used to monitor antiresorptive therapy.
- NTX (N-terminal telopeptide): Another collagen fragment reflecting matrix breakdown; measured in serum or urine.
- TRAP5b (Tartrate-resistant acid phosphatase isoform 5b): An enzyme secreted by active osteoclasts, correlating with cell number rather than matrix degradation per se.
- D-pyridinoline and P-pyridinoline: Crosslinking compounds released during collagen breakdown, typically assessed in urine.
Formation Markers
Bone formation markers indicate osteoblast activity and new matrix synthesis:
- P1NP (Procollagen type I N-terminal propeptide): Byproduct of collagen type I synthesis; sensitive and precise measure of formation.
- Bone-specific alkaline phosphatase (bone ALP): Enzyme expressed on osteoblast surfaces, facilitating mineral deposition.
- Osteocalcin (OC): Noncollagenous protein secreted by osteoblasts, incorporated into bone matrix; small amounts circulate in blood.
- Procollagen type I C-terminal propeptide (P1CP): Less commonly used but similarly reflects collagen synthesis.
Clinical Applications of Bone Turnover Markers
Diagnosis and Monitoring
Biochemical biomarkers of bone turnover have become integral in managing metabolic bone disorders. In osteoporosis, markers help predict fracture risk beyond bone mineral density (BMD) assessments. Elevated resorption markers often precede detectable BMD loss, allowing earlier intervention. During antiresorptive therapy (e.g., bisphosphonates or denosumab), decreases in CTX or NTX validate treatment adherence and efficacy, while increases may signal suboptimal response or secondary causes of bone loss.
In conditions such as Paget’s disease of bone, characterized by accelerated and disorganized remodeling, extremely high levels of bone ALP and other markers confirm disease activity and guide dosing of calcitonin or bisphosphonates. Similarly, bone metastases from breast or prostate cancer manifest with abnormal turnover patterns; rising markers can indicate disease progression or response to targeted therapies.
Challenges and Limitations
Despite their potential, bone turnover markers face several hurdles. Preanalytical variability is significant: circadian rhythms affect marker levels, with CTX typically peaking in early morning and declining throughout the day. Food intake, exercise, and seasonal changes further confound measurements. Inter‐ and intra‐laboratory standardization remains an issue, as different assay platforms yield noncomparable results. Clinicians must consider these factors when interpreting serial measurements.
Additionally, bone turnover markers reflect global skeletal activity, lacking site specificity. A localized fracture or surgical intervention may transiently elevate markers, obscuring underlying metabolic trends. Finally, concurrent illnesses (renal dysfunction, liver disease, inflammatory states) can alter marker clearance or production, limiting clinical utility in complex patients.
Future Perspectives
Ongoing research aims to refine the role of turnover markers in precision medicine. Novel analytes, such as microRNAs and metabolites linked to bone cell differentiation, offer promise as more specific or stable indicators. Integration of markers with fracture risk algorithms and imaging modalities may yield composite scores that outperform current standards. Point-of-care testing and assay harmonization initiatives will enhance accessibility and inter‐center comparability, enabling broader adoption in routine practice.
Personalized therapeutic strategies based on marker profiles could optimize treatment duration, switch timing, and combination regimens. For example, patients exhibiting low formation but high resorption may benefit from anabolic agents followed by potent antiresorptives, whereas others with balanced turnover may require only maintenance therapy. As our understanding deepens, biochemical turnover markers will likely become indispensable in tailoring bone disease management for individual patients.