Innovations in bone repair strategies have paved the way for next-generation therapies that integrate advanced materials science with regenerative medicine. This article explores the development of bioactive materials tailored for enhancing natural bone healing processes. Focusing on design principles, biological mechanisms, and translational challenges, we offer an in-depth analysis of how engineered constructs can facilitate robust tissue regeneration while overcoming limitations of traditional grafts.
Material Design and Composition
Engineering effective bone graft substitutes requires a precise understanding of the physicochemical properties that govern cellular responses. Researchers have prioritized scaffolds that combine mechanical integrity with biological cues, ensuring both load-bearing capacity and active participation in regeneration.
Key Components:
- Hydroxyapatite (HA): Mimics the mineral phase of native bone, providing osteoconductivity and enhancing integration with host tissue.
- Biodegradable Polymers: Materials such as poly(lactic-co-glycolic acid) (PLGA) and polycaprolactone (PCL) tailor degradation rates to match new tissue formation.
- Nanotechnology: Nanofiber matrices and nanoparticles enable controlled presentation of bioactive agents and increase surface area for cell attachment.
- Composite Scaffolds: Hybrid constructs merging ceramics and polymers achieve optimal mechanical strength while maintaining porosity for vascular ingrowth.
Porosity and Mechanical Strength
Porosity is critical for nutrient transport and vascular infiltration, yet increasing pore volume often compromises mechanical stability. By employing techniques like 3D printing and gas foaming, engineers can fabricate hierarchical pore structures that balance compressive strength with interconnected channels. Advanced modeling tools predict stress distribution, enabling custom designs for load-bearing bones such as femoral or tibial segments.
Bioactive Coatings and Surface Functionalization
Surface modifications play a pivotal role in modulating cell-material interactions. Coatings loaded with growth factors such as bone morphogenetic proteins (BMPs) or peptides like RGD sequences enhance cellular adhesion and guide differentiation. Plasma treatments and silanization strategies introduce reactive groups, promoting the covalent attachment of signaling molecules that regulate osteogenesis.
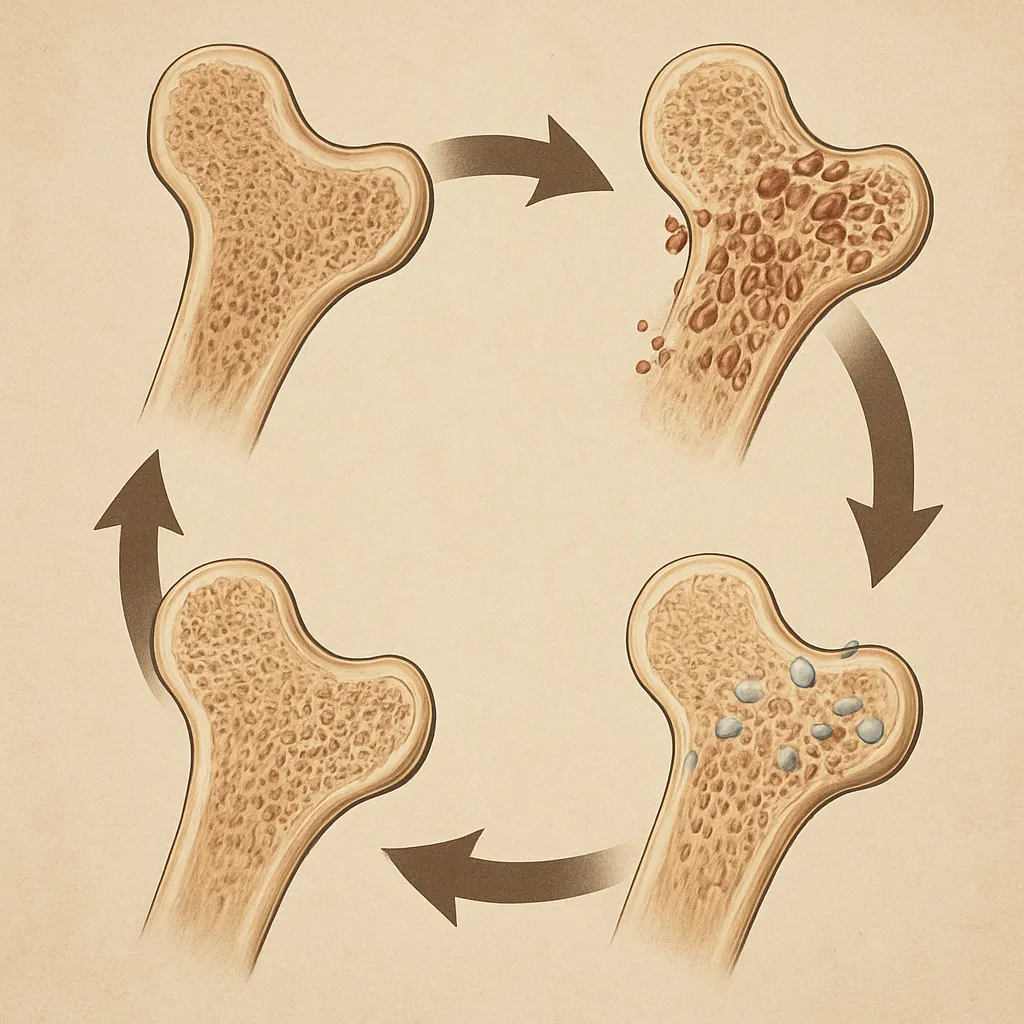
Mechanisms of Action in Bone Regeneration
Successful bone healing depends on orchestrating multiple biological events: inflammation, cell recruitment, matrix deposition, and remodeling. Bioactive materials can actively engage each phase by providing structural support and delivering biochemical cues.
Modulation of Inflammatory Response
An initial inflammatory phase is essential to clear debris and recruit progenitor cells. However, prolonged inflammation can impair healing. Materials incorporating anti-inflammatory agents or designed to evade excessive immune activation encourage a balanced macrophage response. M2-polarizing cues, such as interleukin-4 (IL-4) release, have shown promise in accelerating transition to the reparative stage.
Stem Cell Recruitment and Differentiation
Localized delivery of chemoattractants like stromal cell-derived factor-1 (SDF-1) recruits endogenous stem cells to the defect site. Once present, these multipotent cells require lineage-specific signals. Scaffolds enriched with transforming growth factor-beta (TGF-β) or BMP-2 drive mesenchymal stem cells toward osteoblastic fate, laying down new matrix components.
Angiogenesis and Vascular Integration
New bone formation is intimately linked with blood vessel growth. Strategies to promote angiogenesis include embedding vascular endothelial growth factor (VEGF) within biodegradable microspheres or constructing channel networks that guide capillary sprouting. Co-culture systems combining endothelial cells with osteoprogenitors have produced pre-vascularized constructs that enhance in vivo engraftment.
Controlled Drug Delivery
Drug-eluting scaffolds can release antibiotics, anti-resorptive drugs, or nucleic acids in a sustained manner. Nanoparticles and microparticles loaded with therapeutic agents are integrated into the matrix to prevent infection, inhibit osteoclast activity, or introduce gene therapy vectors. This integrated drug delivery approach reduces systemic side effects and concentrates treatment at the injury site.
Clinical Applications and Future Directions
Translating benchside innovations to clinical reality requires rigorous evaluation of safety, efficacy, and manufacturability. Recent trials have begun to demonstrate the potential of bioactive materials in complex bone defects, spinal fusion, and dental implants.
Emerging Trends:
- Personalized Medicine: Patient-specific implants derived from imaging data ensure anatomical fit and optimal load distribution.
- Smart Materials: Stimuli-responsive polymers that adjust their properties in response to pH, temperature, or mechanical stress enable dynamic adaptation during healing.
- Gene-Activated Scaffolds: Incorporation of plasmid DNA or viral vectors within the material unlocks sustained in situ production of therapeutic proteins.
- In Situ Bioprinting: Advanced surgical robots can deposit cell-laden bioinks directly into defects, forming living constructs that integrate immediately with surrounding tissue.
Regulatory and Manufacturing Challenges
Ensuring consistent quality across batches, sterilization without compromising bioactivity, and scaling up production remain significant hurdles. Regulatory agencies require comprehensive data on biocompatibility, biodegradation profiles, and long-term outcomes. Collaboration between material scientists, biologists, and clinicians is essential to navigate approval pathways and establish standardized testing protocols.
Future Perspectives
Interdisciplinary research continues to drive the field toward multifunctional platforms that combine mechanical support, biological stimulation, and diagnostic capabilities. Integration of sensors within scaffolds could enable real-time monitoring of pH, mechanical strain, or inflammatory markers. Moreover, leveraging artificial intelligence to analyze patient data may facilitate the design of tailored constructs with optimized performance.
By harnessing the synergy of advanced materials, molecular biology, and clinical expertise, the next generation of bone healing therapies promises to restore function more effectively and reduce recovery times, offering new hope for patients suffering from traumatic injuries, degenerative diseases, or congenital defects.