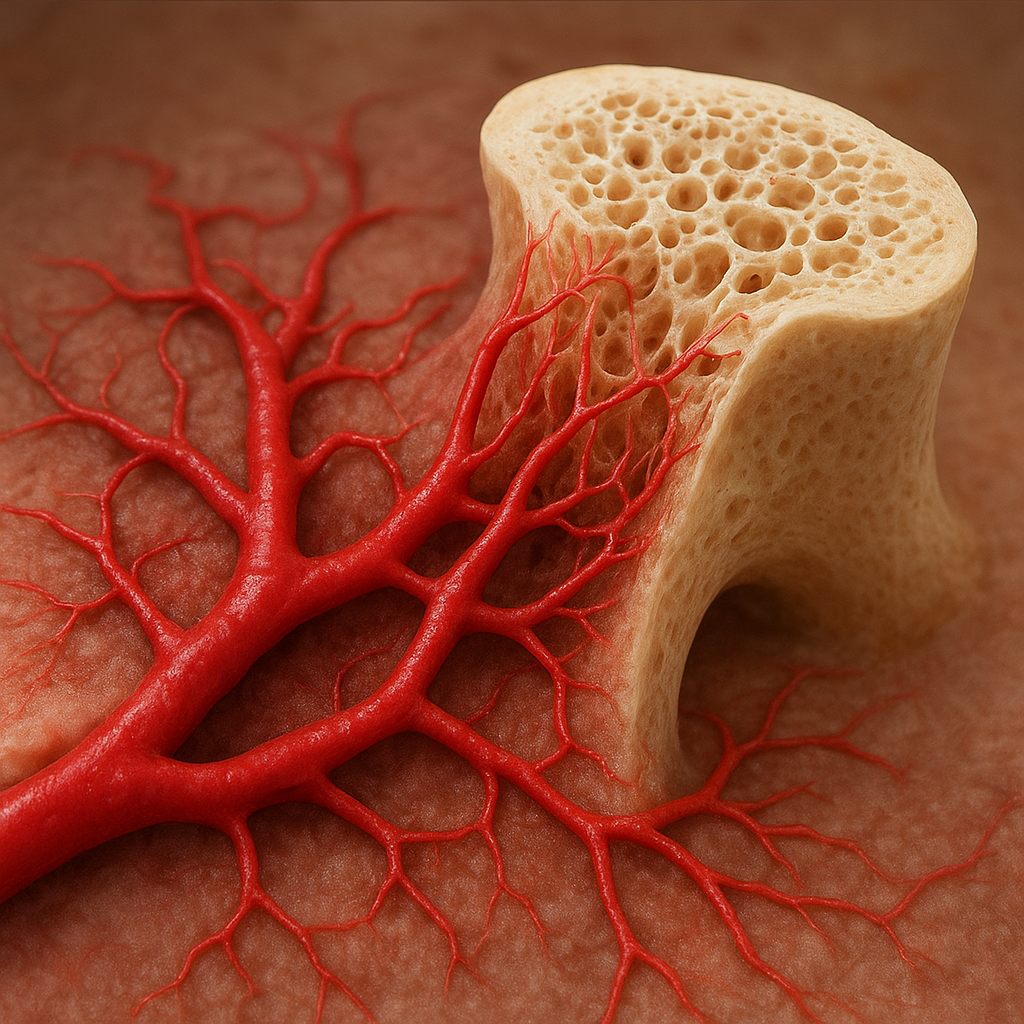
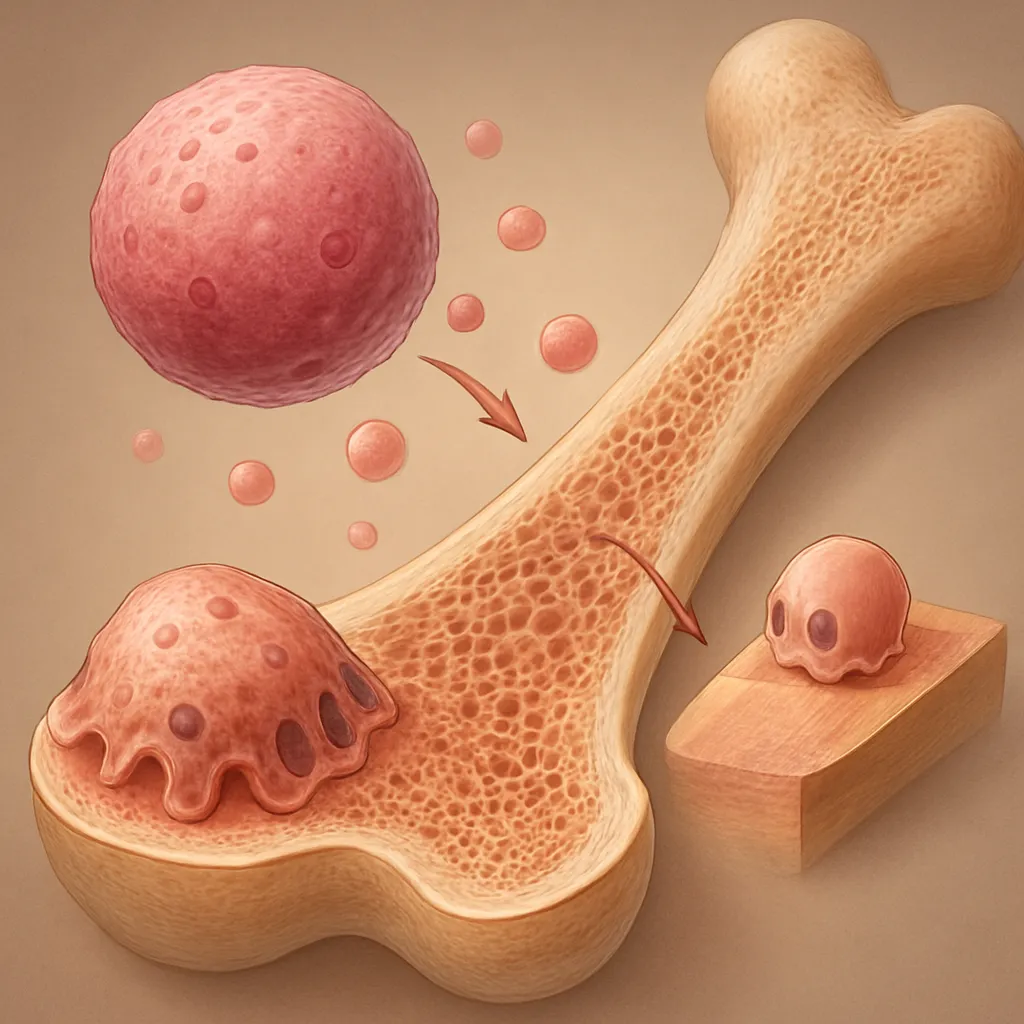
Bone regeneration represents a complex interplay between cellular events and vascular supply. Adequate blood vessel formation is essential for delivering nutrients, oxygen, and signaling molecules to the repair site. This article explores the pivotal role of angiogenesis in coordinating the biological processes underlying bone healing and highlights innovative strategies to enhance skeletal repair.
Angiogenesis and Bone Healing Mechanisms
Following a fracture or bone defect, a well-orchestrated cascade of events is initiated. The initial inflammatory phase recruits immune cells that not only clear debris but also release cytokines to stimulate vessel sprouting. Without timely neovascular invasion, oxygen deprivation impairs cellular functions, delaying or arresting new bone formation.
Phases of Bone Healing
The reparative response unfolds in distinct stages:
- Inflammation: Recruitment of macrophages and neutrophils to the injury site; establishment of a fibrin-rich clot.
- Soft Callus Formation: Proliferation of chondrocytes creates a cartilaginous scaffold; emerging capillaries infiltrate the matrix.
- Hard Callus Formation: Replacement of cartilage with woven bone through osteogenesis; robust vascular networks support mineral deposition.
- Remodeling: Transition of woven bone into lamellar bone; restoration of original architecture and mechanical strength.
Interdependence of Vessels and Bone Cells
Endothelial cells form the lining of new capillaries and secrete factors that promote osteoblast differentiation. Conversely, osteoblasts produce pro-angiogenic mediators to sustain vessel growth. This crosstalk ensures synchronized development of bone and its nutrient supply.
Growth Factors and Cellular Players
Key molecular signals govern the sprouting and maturation of blood vessels within regenerating bone. Understanding these factors enables targeted enhancement of repair processes.
Pro-Angiogenic Mediators
- VEGF (Vascular Endothelial Growth Factor): Stimulates proliferation and migration of endothelial cells.
- FGF (Fibroblast Growth Factor): Promotes endothelial cell survival and matrix remodeling.
- PDGF (Platelet-Derived Growth Factor): Recruits pericytes and smooth muscle cells for vessel stabilization.
- Angiopoietins: Regulate endothelial cell sprouting and vessel maturation phases.
Endothelial Cells and Pericytes
Endothelial progenitors invade hypoxic bone callus regions, guided by chemotactic gradients. Pericytes wrap around nascent vessels, ensuring structural integrity and controlling permeability. Together, they form a functional microcirculation capable of sustaining active bone formation.
Stem Cells and Progenitors
Mesenchymal stem cells (MSCs) residing in the periosteum and bone marrow differentiate into osteoblasts under the influence of both mechanical cues and vascular signals. Their secretome includes pro-angiogenic cytokines that further amplify vessel invasion. The interplay between mesenchymal progenitors and endothelial populations is central to balanced tissue regeneration.
Therapeutic Approaches Targeting Angiogenesis
Translational strategies aim to accelerate vascular ingrowth and improve clinical outcomes in bone defects. Innovative techniques harness biological, material, and genetic tools to modulate the local microenvironment.
Biomaterial-based Strategies
Engineered scaffolds serve as templates for cell attachment and vessel infiltration. Key design features include:
- Porosity and interconnectivity to facilitate capillary penetration.
- Incorporation of scaffolds embedded with growth factor reservoirs for sustained release.
- Use of biomaterials such as collagen, hydroxyapatite, and bioceramics that mimic native extracellular matrix.
- Surface modification with peptides (e.g., RGD motifs) to enhance endothelial adhesion.
Gene and Cell Therapy
Local delivery of genes encoding VEGF or other angiogenic factors can induce prolonged expression at the defect site. Alternatively, transplantation of endothelial progenitor cells or genetically modified MSCs holds promise for synergistic vascular and bone tissue regeneration.
Challenges and Future Directions
Despite encouraging preclinical data, several obstacles limit the widespread adoption of pro-angiogenic therapies in orthopedics.
Translational Barriers
- Host comorbidities such as diabetes and smoking impair vascular responsiveness and healing capacity.
- Regulatory hurdles for cell and gene-based products prolong clinical translation timelines.
- Risk of aberrant vessel formation or uncontrolled angiogenesis raising safety concerns.
Emerging Technologies
Advancements in bioprinting allow fabrication of constructs with pre-vascularized networks. Microfluidic platforms enable in vitro modeling of bone-vascular interfaces under controlled shear and oxygen gradients. Integration of sensor technologies may permit real-time monitoring of oxygen tension and perfusion within grafts, optimizing therapeutic interventions.
In summary, targeting the vascular component of bone repair offers a powerful avenue to enhance regenerative outcomes. Continued interdisciplinary research will be essential to overcome current limitations and fully harness the potential of angiogenesis-driven bone regeneration.