Bone loss is a significant complication affecting patients with end-stage renal disease undergoing dialysis. Chronic kidney disease disrupts the balance of minerals and hormones essential for bone health, leading to a spectrum of disorders collectively termed renal osteodystrophy. Understanding the underlying mechanisms, risk factors, and management strategies is crucial to mitigate the elevated fracture risk and improve quality of life in this vulnerable population.
Pathophysiology of Bone Loss in Dialysis Patients
The kidneys play a central role in maintaining calcium homeostasis and regulating phosphate excretion. Progressive renal impairment leads to **phosphate retention**, decreased activation of vitamin D, and disturbances in mineral metabolism. As serum phosphate rises, levels of parathyroid hormone (PTH) increase, causing secondary hyperparathyroidism and accelerated bone turnover.
- Vitamin D deficiency: Reduced renal 1α-hydroxylase activity limits conversion of 25(OH)D to active 1,25(OH)2D, impairing calcium absorption and skeletal mineralization.
- Secondary hyperparathyroidism: Chronically elevated PTH stimulates osteoclast-mediated bone resorption, leading to cortical bone loss and trabecular thinning.
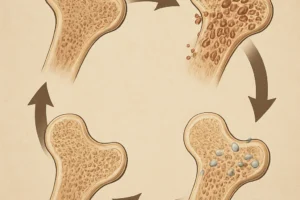
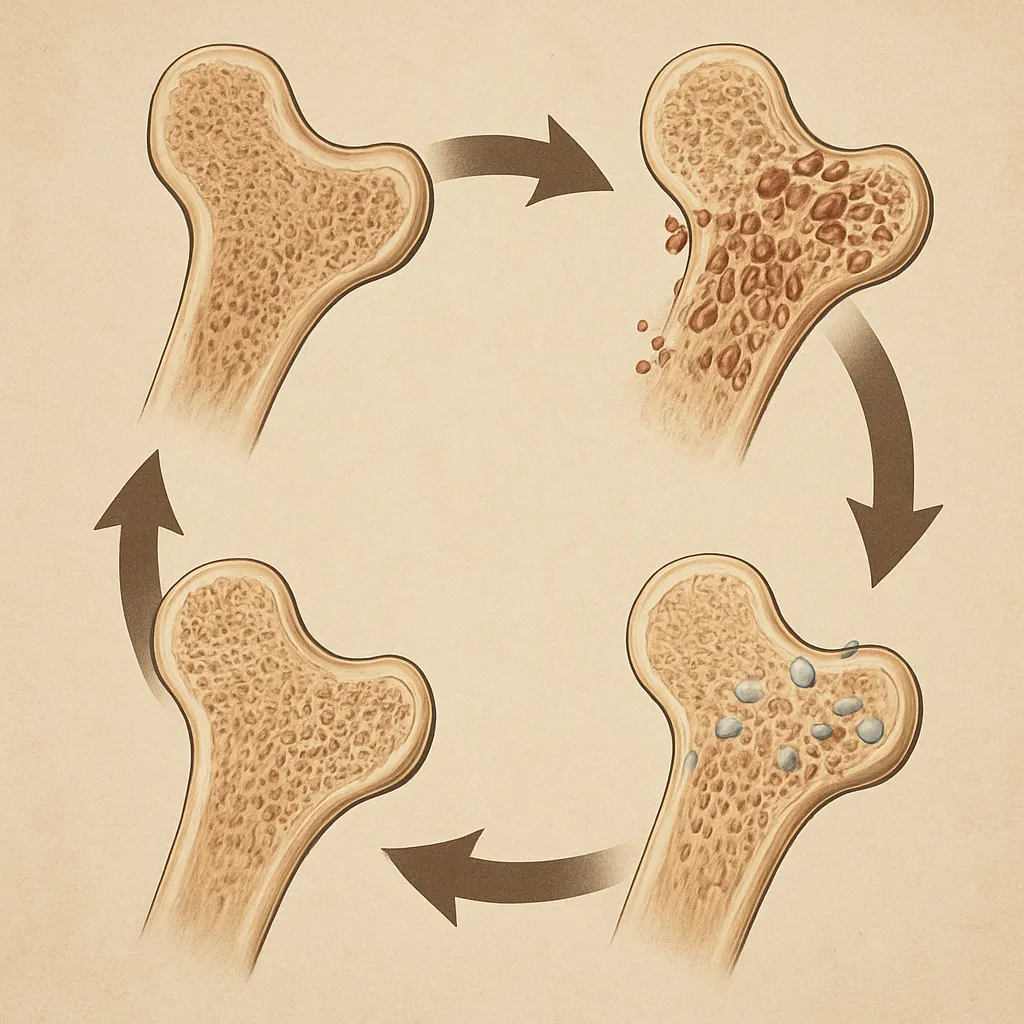
- Renal osteodystrophy: A collective term for bone disorders ranging from high-turnover lesions to adynamic bone disease characterized by low bone formation.
Persistent imbalance triggers remodeling abnormalities, compromises structural integrity, and reduces bone mineral density, rendering bones more susceptible to microfractures.
Risk Factors and Clinical Implications
Multiple factors contribute to the severity of bone loss in dialysis patients, often acting synergistically:
Metabolic and Hormonal Contributors
- Elevated serum phosphate and PTH levels accelerating bone turnover.
- Hypocalcemia due to reduced vitamin D activation.
- Chronic inflammation and oxidative stress impairing osteoblast function.
Treatment-Related Factors
- Inadequate dialysis adequacy leading to persistent uremic toxins that interfere with bone cell activity.
- Use of aluminum-based phosphate binders causing osteomalacia.
- Excessive calcium-based binders inducing adynamic bone disease.
Clinical Consequences
Bone fragility in dialysis patients often presents with:
- Spontaneous fractures of the hip, wrist, and vertebrae.
- Chronic bone pain and reduced mobility.
- Increased morbidity and mortality due to complications.
Diagnostic Approaches and Biomarkers
A comprehensive assessment integrates biochemical markers, imaging, and sometimes bone biopsy:
- Serum measurements: phosphate, calcium, PTH, alkaline phosphatase, and 25(OH)D levels.
- Dual-energy X-ray absorptiometry (DEXA): Evaluates bone mineral density at critical sites.
- Bone histomorphometry: Gold standard for distinguishing high-turnover from adynamic disease, though invasive.
- Emerging biomarkers: Fibroblast growth factor-23 (FGF-23) and sclerostin as indicators of mineral metabolism disorders.
Early identification of high-risk patients through routine monitoring allows timely intervention to preserve skeletal health.
Management Strategies and Therapeutic Interventions
An integrated approach targeting the underlying pathologies can stabilize or reverse bone loss:
- Dietary phosphate restriction combined with phosphate binders to control hyperphosphatemia.
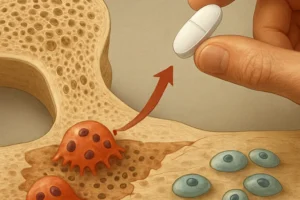
- Administration of active vitamin D analogs (calcitriol, paricalcitol) to suppress excessive PTH secretion and enhance calcium absorption.
- Calcimimetics (cinacalcet) to modulate the calcium-sensing receptor and lower PTH levels without inducing hypercalcemia.
- Transition to newer non-calcium phosphate binders (sevelamer, lanthanum) to avoid calcium overload and adynamic bone disease.
Adjunctive Measures
- Physical activity and weight-bearing exercises to stimulate bone formation and improve muscle strength.
- Correction of metabolic acidosis to reduce bone buffering demands.
- Individualized dialysis prescriptions optimizing removal of uremic inhibitors of bone turnover.
Future Directions
Novel therapies under investigation include monoclonal antibodies against sclerostin to promote bone formation, and modulators of Wnt signaling pathways. Precision medicine approaches aim to tailor treatment based on genetic and biochemical profiles, maximizing efficacy while minimizing adverse effects.