The extracellular matrix of bone represents a complex and dynamic network, essential for providing structural integrity and enabling a variety of biological processes. In-depth understanding of its molecular composition and functional roles is crucial for advancements in orthopedics, tissue engineering, and the treatment of metabolic bone diseases. This article explores the biochemical constituents of bone matrix, elucidates how these components interact to sustain mechanical strength and cellular activity, and examines clinical implications in health and disease.
Bone Matrix Composition
The bone matrix is comprised of two primary phases: an organic component that confers flexibility and toughness, and an inorganic phase that delivers mechanical rigidity. The coordinated interplay between these phases underlies the unique properties of bone tissue.
Organic Phase
- Collagen Type I: Constitutes approximately 90% of the organic matrix. Tropocollagen molecules align in a quarter-staggered pattern, forming fibrils that resist tensile forces.
- Non-Collagenous Proteins: Include osteocalcin, osteopontin, bone sialoprotein, and others. These proteins regulate mineral nucleation, cell–matrix interactions, and remodeling processes.
- Proteoglycans and Glycoproteins: Decorin, biglycan, and fibronectin participate in matrix organization and growth factor binding, influencing osteoblast proliferation and differentiation.
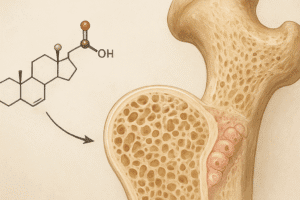
Inorganic Phase
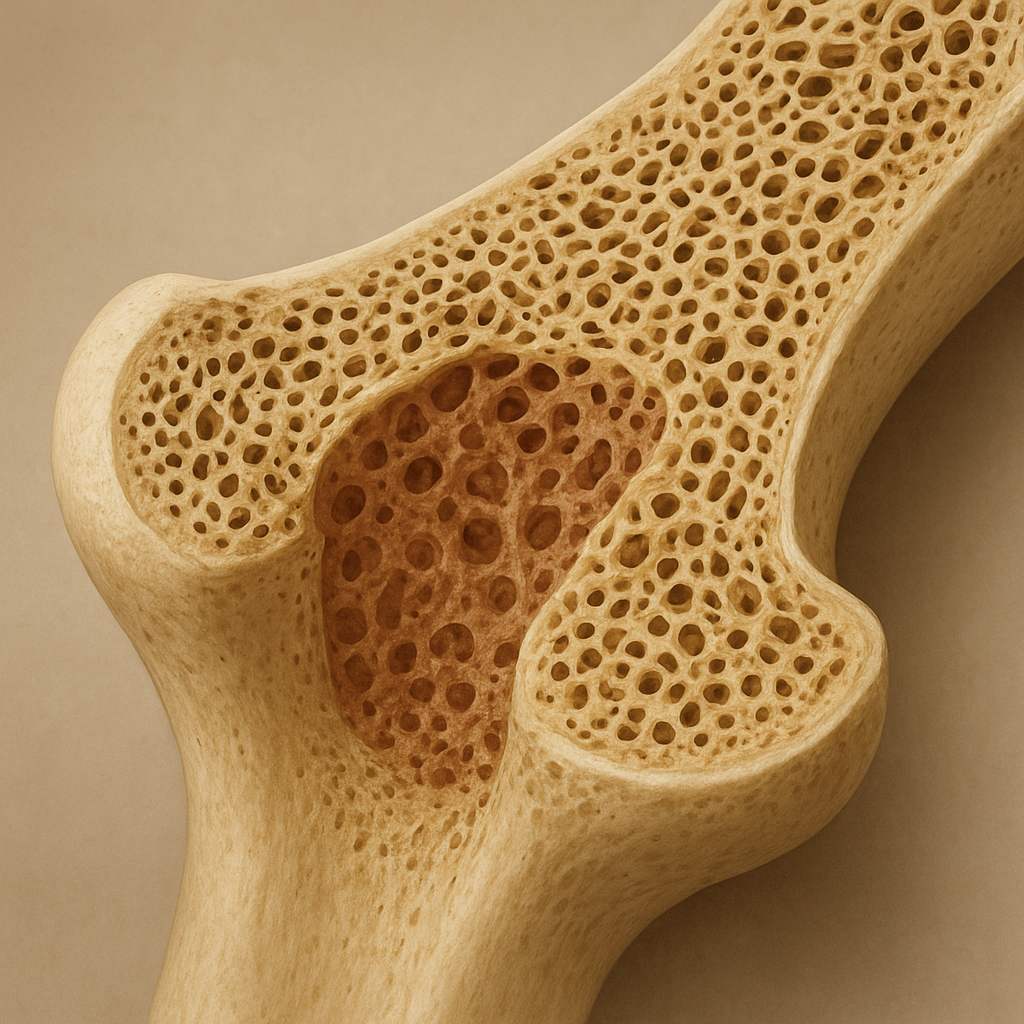
The mineral fraction, predominantly composed of hydroxyapatite crystals [Ca10(PO4)6(OH)2], accounts for about 65% of the bone’s mass. These nanometer-scale platelets are deposited within and between collagen fibrils in a highly regulated process known as mineralization. The precise orientation and size of crystals determine the bone’s stiffness and compressive strength.
Structural Organization and Mechanical Function
Bone tissue is organized at multiple hierarchical levels to optimally distribute loads and resist fracture. From the molecular arrangement of collagen-mineral composites to the macroscopic architecture of trabecular and cortical bone, each scale confers specific functional advantages.
Molecular and Fibrillar Level
- Collagen Fibrils Mineralized with Hydroxyapatite: Mineral platelets align along the long axis of fibrils, providing a balance of tensile and compressive strength.
- Cross-linking: Enzymatic and non-enzymatic cross-links enhance fibril stability and resistance to enzymatic degradation.
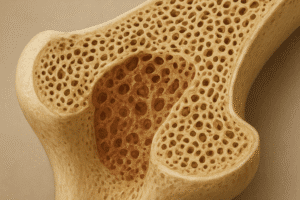
Microstructural Level
- Trabecular (Spongy) Bone: A porous, honeycomb-like network that absorbs shock and distributes loads. Its high surface area supports active remodeling and ion exchange.
- Cortical (Compact) Bone: Dense and organized into osteons—cylindrical structures with central Haversian canals. Provides the majority of bone’s bending and torsional strength.
Macroscopic Level
Long bones display a tubular structure with thicker cortical walls at the diaphysis and expanded trabecular-rich metaphyses near joints. This design leverages geometry to optimize weight-to-strength ratio, allowing efficient movement and load bearing during locomotion.
Cellular Interactions and Remodeling Dynamics
Bone is a living tissue maintained by the coordinated actions of osteoblasts, osteoclasts, and osteocytes. The matrix not only supports these cells but also actively regulates their behavior through matrix-derived signals.
Osteoblasts and Matrix Synthesis
- Matrix Secretion: Osteoblasts synthesize collagen and non-collagenous proteins, organizing them into a scaffold for mineral deposition.
- Vesicular Mineralization: Matrix vesicles loaded with calcium and phosphate ions initiate hydroxyapatite crystal formation.
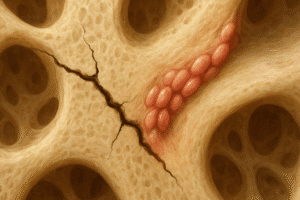
Osteoclasts and Matrix Resorption
- Acidification and Proteolysis: Osteoclasts secrete protons and proteolytic enzymes (e.g., cathepsin K) to dissolve mineral and degrade organic matrix.
- Bone Turnover: Balanced resorption and formation cycles ensure renewal, adaptation to mechanical loads, and maintenance of mineral homeostasis.
Osteocytes and Mechanotransduction
Embedded within the mineralized matrix, osteocytes extend dendritic processes through canaliculi, sensing mechanical strain and orchestrating remodeling by releasing signaling molecules such as sclerostin and RANKL.
Clinical Significance and Applications
Alterations in bone matrix composition or organization contribute to various pathologies. Understanding these changes informs both diagnostic strategies and the development of novel therapies.
Metabolic Bone Diseases
- Osteoporosis: Characterized by reduced bone mass and microarchitectural deterioration. Diminished collagen quality and mineral density increase fracture risk.
- Osteogenesis Imperfecta: A genetic defect in collagen production leads to brittle bones. Mutations in COL1A1 or COL1A2 genes impair fibril assembly.
Biomaterials and Tissue Engineering
Advances in scaffold design seek to mimic native bone matrix properties. Composite materials combine biocompatible polymers (e.g., polycaprolactone) with bioactive ceramics (biomaterials such as synthetic hydroxyapatite) to promote osteointegration and guide tissue regeneration.
- 3D-Printed Scaffolds: Tailored pore architecture supports vascularization and cell infiltration.
- Growth Factor Delivery: Incorporation of BMPs (bone morphogenetic proteins) enhances osteoinduction within a scaffold matrix.
Diagnostic Imaging and Biomarkers
Quantitative techniques—such as dual-energy X-ray absorptiometry (DXA) and high-resolution peripheral quantitative computed tomography (HR-pQCT)—assess bone mineral density and microarchitecture. Circulating markers like alkaline phosphatase, C-terminal telopeptide, and proteoglycans fragments reflect matrix turnover rates.
Emerging Research Trends
Ongoing investigations target the fine-tuning of matrix composition to enhance bone repair and combat degenerative conditions.
- Nanotechnology: Engineering of nano-scale hydroxyapatite coatings to improve implant integration and mechanical compatibility.
- Gene Editing: CRISPR/Cas9 approaches aim to correct mutations in collagen genes or modulate expression of key matrix regulators.
- Mechanobiology: Elucidation of how mechanical cues influence matrix deposition paves the way for rehabilitation protocols that optimize bone health.