Bone marrow-derived stem cells have emerged as a cornerstone in regenerative medicine, particularly within the realm of orthopedic interventions. These cells possess unique properties that enable them to repair, regenerate, and restore damaged musculoskeletal tissues. As researchers continue to unveil the complexities of cellular behavior, the clinical potential of these cells in treating fractures, cartilage defects, and degenerative bone diseases grows exponentially.
Principles of Bone Marrow-Derived Stem Cells
Bone marrow is home to two primary stem cell populations: hematopoietic stem cells (HSCs) and mesenchymal stem cells (MSCs). While HSCs give rise to blood components, MSCs are multipotent progenitors capable of differentiating into osteoblasts, chondrocytes, and adipocytes. This multipotent nature underpins their versatility in orthopedic applications.
Cellular Origin and Niche
- HSCs reside in specialized niches regulated by stromal cells, growth factors, and extracellular matrix interactions.
- MSCs are found around blood vessels within the marrow, where they respond to mechanical cues, oxygen tension, and signaling cytokines.
- The dynamic crosstalk between MSCs and HSCs influences hematopoiesis and tissue homeostasis.
Key Functional Properties
- Proliferation: MSCs display robust expansion capacity in vitro, essential for generating therapeutic cell doses.
- Differentiation: Under appropriate culture conditions, they divert toward osteogenic or chondrogenic lineages, offering targeted repair strategies.
- Immunomodulation: These cells secrete anti-inflammatory mediators and modulate T-cell activity, enhancing graft survival and reducing rejection.
Isolation and Characterization Techniques
Successful therapeutic use hinges on reliable isolation and precise characterization of marrow-derived stem cells. Standardized protocols ensure reproducibility and safety.
Harvesting Procedures
- Bone marrow aspiration is typically performed from the iliac crest under local or general anesthesia.
- Volume aspirated ranges from 20 to 60 milliliters per site to optimize MSC yield without excessive blood dilution.
Cell Separation and Enrichment
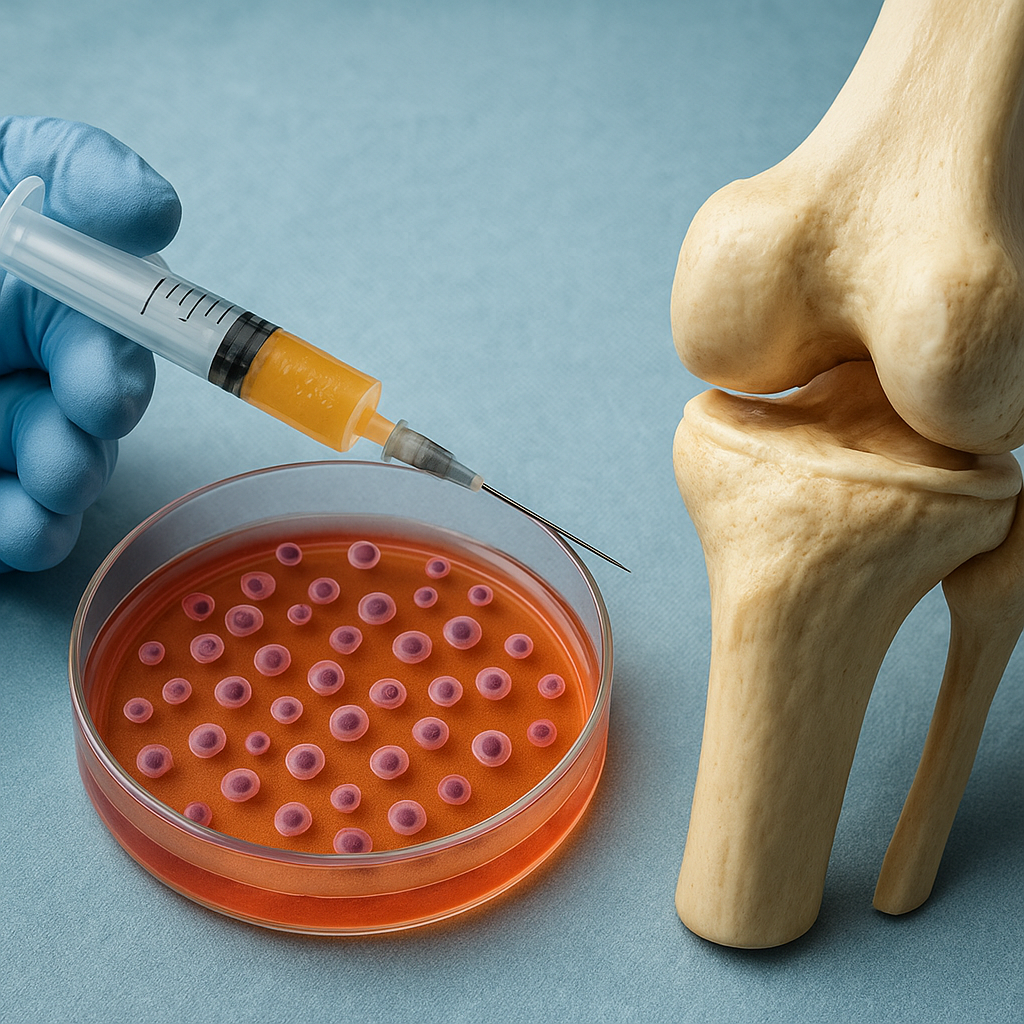
- Density-gradient centrifugation isolates mononuclear cells, enriching the stem cell fraction.
- Magnetic-activated cell sorting (MACS) or fluorescence-activated cell sorting (FACS) further purifies MSC populations using surface markers like CD73, CD90, and CD105.
Quality Control and Characterization
- Phenotypic analysis confirms the absence of hematopoietic markers such as CD34 and CD45.
- Functional assays evaluate differentiation potential into osteogenic, chondrogenic, and adipogenic lineages.
- Genetic stability is assessed via karyotyping to prevent malignant transformation during ex vivo expansion.
Applications in Orthopedic Surgery
Bone marrow-derived stem cells have revolutionized treatment paradigms across various orthopedic conditions by leveraging their inherent capacity for regeneration and tissue remodeling.
Fracture Repair
- Nonunion fractures benefit from MSC-enriched grafts that release osteoinductive factors, accelerating callus formation and consolidation.
- Combining MSCs with biocompatible scaffold materials allows for precise defect filling and mechanical support.
Articular Cartilage Regeneration
- Osteochondral defects treated with MSC-laden hydrogels show improved integration and hyaline-like cartilage restoration.
- Preconditioning MSCs in chondrogenic media enhances secretion of matrix proteins such as collagen II and aggrecan.
Spinal Fusion and Intervertebral Disc Repair
- MSC-based therapies reduce the need for autologous bone grafts, minimizing donor site morbidity.
- Intravertebral administration aims to improve disc hydration and retard degenerative changes by modulating inflammatory mediators.
Osteonecrosis Management
- Core decompression combined with MSC injection into necrotic lesions promotes neovascularization and prevents femoral head collapse.
Challenges and Future Directions
Despite promising outcomes, several obstacles must be addressed to translate bone marrow-derived stem cell therapies into widespread clinical use.
Scalability and Manufacturing
- Producing clinical-grade MSCs in compliance with good manufacturing practices (GMP) is resource-intensive.
- Developing bioreactor systems can enhance proliferation rates while maintaining phenotypic integrity.
Engraftment and Survival
- Ensuring long-term engraftment requires improvement of cell delivery methods and creation of bioactive scaffold environments.
- Hypoxia-preconditioning and genetic modification techniques may bolster cell survival post-implantation.
Regulatory and Ethical Considerations
- Harmonizing global regulations on stem cell therapies is necessary to streamline clinical trials.
- Ethical sourcing of bone marrow, informed consent, and patient safety remain paramount concerns.
Emerging Innovations
- 3D bioprinting of MSC-laden constructs offers customized implants for complex orthopedic defects.
- Combining MSCs with gene therapy aims to deliver targeted growth factors for enhanced differentiation and tissue integration.
- Exosome-based treatments harness the paracrine activity of MSCs without the risks associated with cellular transplantation.