The complex interplay between the immune system and skeletal homeostasis in rheumatoid arthritis patients reveals critical insights into disrupted bone health. This article examines the mechanisms of bone remodeling, the clinical impact of bone loss, and potential therapeutic approaches to preserve skeletal integrity in individuals affected by rheumatoid arthritis.
Mechanisms of Altered Bone Remodeling
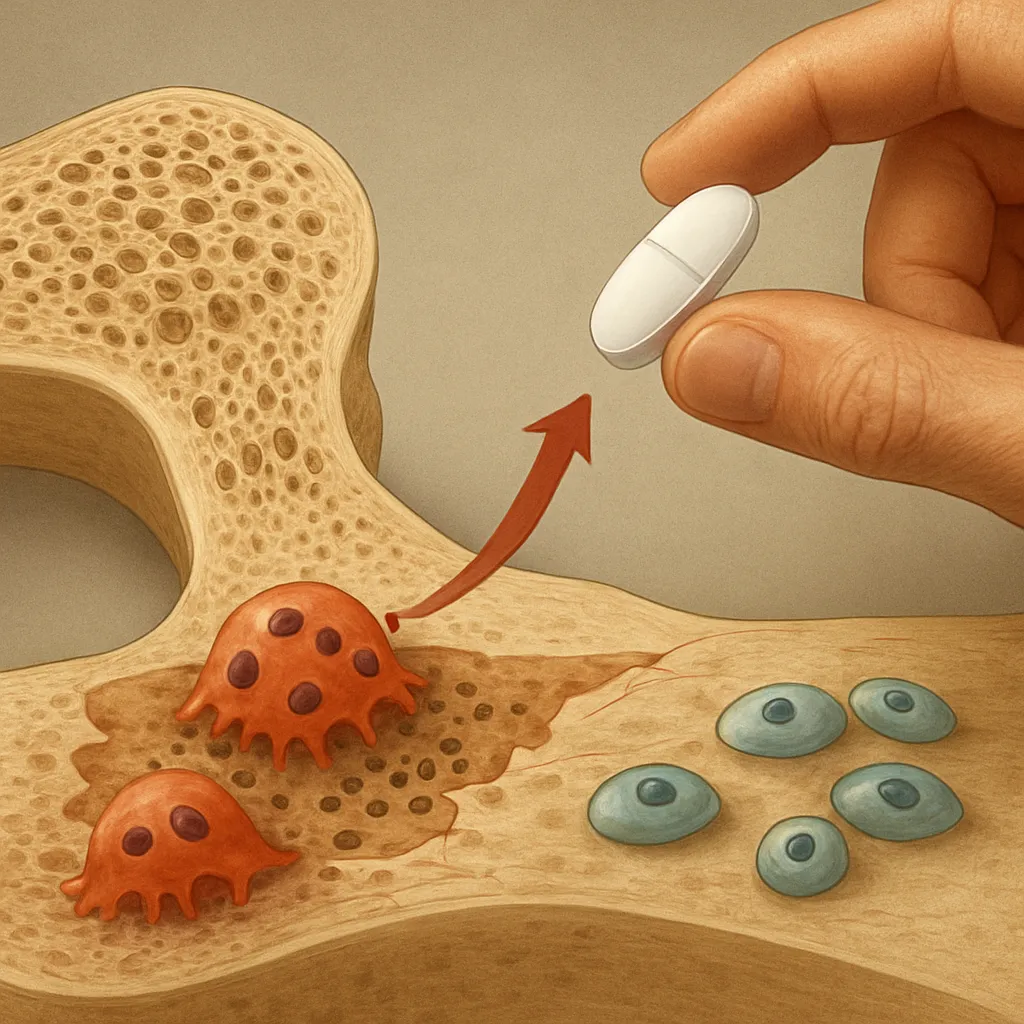
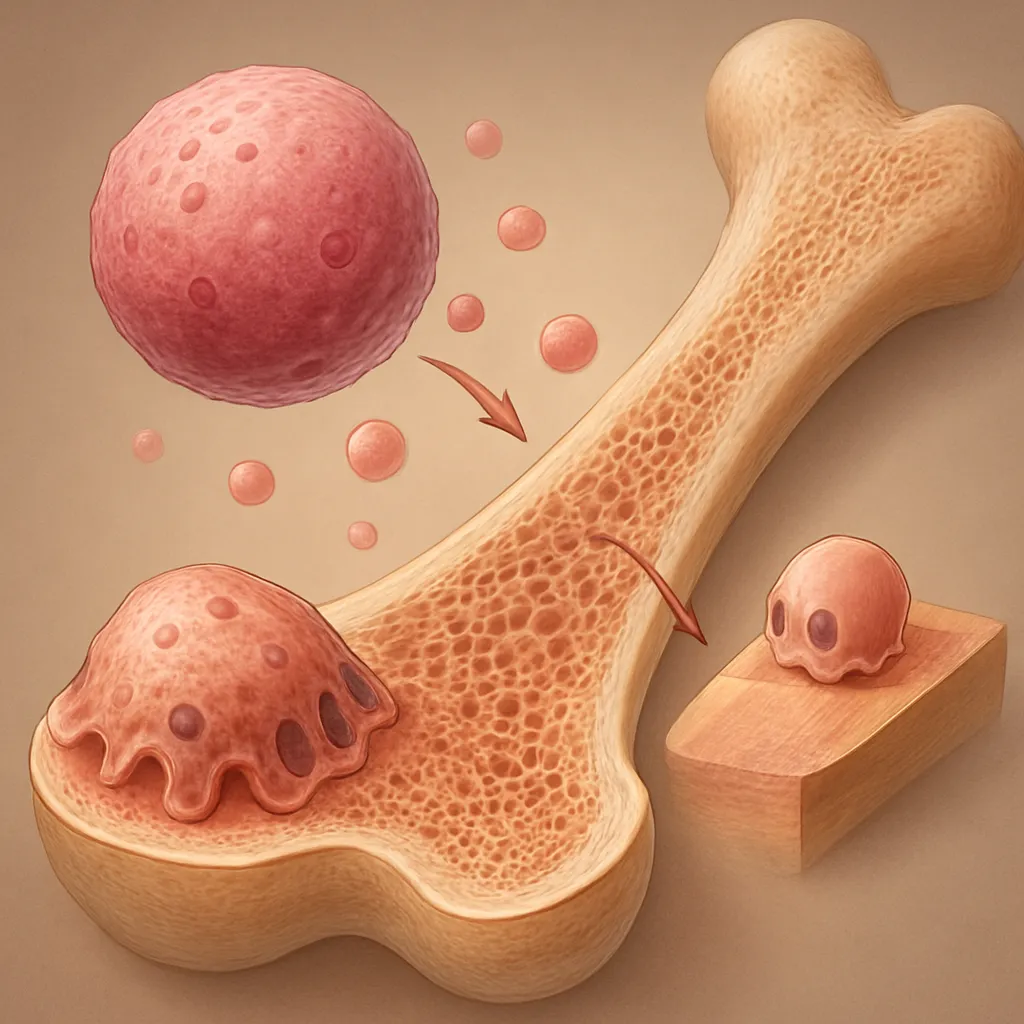
In rheumatoid arthritis, persistent inflammation drives a cascade of cellular events that shift bone balance toward resorption. Synovial macrophages and T cells produce high levels of pro-resorptive cytokines such as tumor necrosis factor-alpha (TNF-α), interleukin-1 (IL-1), and interleukin-6 (IL-6). These factors promote differentiation and activation of osteoclasts via the RANK/RANKL/OPG signaling axis. Specialized stromal cells and activated T lymphocytes upregulate receptor activator of nuclear factor kappa B ligand (RANKL), while downregulating osteoprotegerin (OPG), a decoy receptor that normally inhibits osteoclastogenesis. The resulting imbalance accelerates cortical and trabecular bone loss.
Beyond cytokine-driven osteoclast activation, autoantibodies such as anti-citrullinated protein antibodies (ACPAs) exert a direct pro-osteoclastogenic effect, further enhancing local and systemic bone erosion. Additionally, chronic inflammation impairs osteoblast function through Wnt pathway antagonism, reducing bone formation and mineralization.
Clinical Implications and Assessment
Patients with rheumatoid arthritis exhibit significant reductions in bone density compared to age-matched controls, leading to an elevated risk of fragility fractures. Both peripheral and axial sites are affected, with vertebral and hip fractures posing particularly high morbidity. Subclinical bone damage can progress even in early disease stages, underscoring the importance of timely assessment.
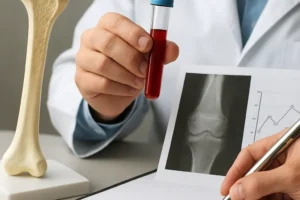
- Dual-energy X-ray absorptiometry (DXA) remains the gold standard for evaluating bone mineral density, although it may underestimate microarchitectural deterioration.
- High-resolution peripheral quantitative computed tomography (HR-pQCT) can capture changes in trabecular thickness and separation, revealing compromised microarchitecture that precedes overt density loss.
- Serum biomarkers such as C-terminal telopeptide (CTX) and procollagen type I N-propeptide (P1NP) provide dynamic insight into bone turnover rates.
Clinicians must integrate disease activity scores, glucocorticoid exposure history, and traditional osteoporosis risk factors to stratify fracture risk accurately. Regular bone health monitoring is essential even in patients achieving clinical remission, as persistent subclinical inflammation can silently perpetuate bone damage.
Therapeutic Strategies for Bone Preservation
Effective management combines targeted suppression of joint inflammation with antiresorptive and anabolic bone agents. Conventional and biological disease-modifying antirheumatic drugs (DMARDs) address underlying autoimmunity while indirectly supporting bone health.
Biologics and Small Molecule DMARDs
Anti-TNF agents and IL-6 receptor inhibitors reduce inflammatory cytokine levels, decreasing osteoclast activation. Studies demonstrate stabilization or modest gains in bone mass in patients receiving tumor necrosis factor blockers. Janus kinase (JAK) inhibitors further modulate cytokine signaling, offering additional protection against bone loss.
Antiresorptive and Anabolic Therapies
Bisphosphonates and denosumab directly inhibit osteoclast-mediated bone resorption, proving effective in rheumatoid arthritis–associated osteoporosis. Denosumab, a monoclonal antibody against RANKL, specifically counters the cytokine-driven osteoclastogenesis prevalent in this population. Meanwhile, teriparatide—a recombinant parathyroid hormone analog—stimulates osteoblast activity, presenting an anabolic option for severe cases.
Adjunctive Nutritional and Lifestyle Interventions
- Ensuring adequate intake of vitamin D and calcium supports mineralization and may modulate immune cell function.
- Weight-bearing exercises and muscle strengthening improve bone strength and reduce fall risk.
- Smoking cessation and moderation of alcohol intake further diminish bone resorption factors.
The judicious use of glucocorticoids remains a therapeutic challenge: while essential for short-term control of flare-ups, long-term exposure exacerbates bone loss through multiple mechanisms. Tapering to the lowest effective dose and initiating bone‐protective agents concurrently is recommended.
Integrating Bone Health into Rheumatoid Arthritis Management
Recognition of bone metabolism disturbances as a core component of rheumatoid arthritis necessitates a multidisciplinary approach. Rheumatologists, endocrinologists, physiotherapists, and primary care providers must collaborate to implement personalized strategies. Monitoring protocols should encompass both inflammatory control and bone quality assessment, ensuring that skeletal preservation is prioritized alongside joint function and patient quality of life.