Bone regeneration in diabetic patients presents a significant clinical challenge due to the complex interplay between systemic metabolic disturbances and local tissue repair mechanisms. Chronic elevations in blood glucose levels disrupt normal cellular functions, impairing the capacity of bone to undergo effective remodeling and repair. This article examines key aspects of bone healing in the context of diabetes mellitus, analyzes the underlying pathophysiological mechanisms, reviews current treatment modalities, and highlights novel strategies that may enhance regenerative outcomes in this high-risk population.
Background on Bone Remodeling and Diabetes
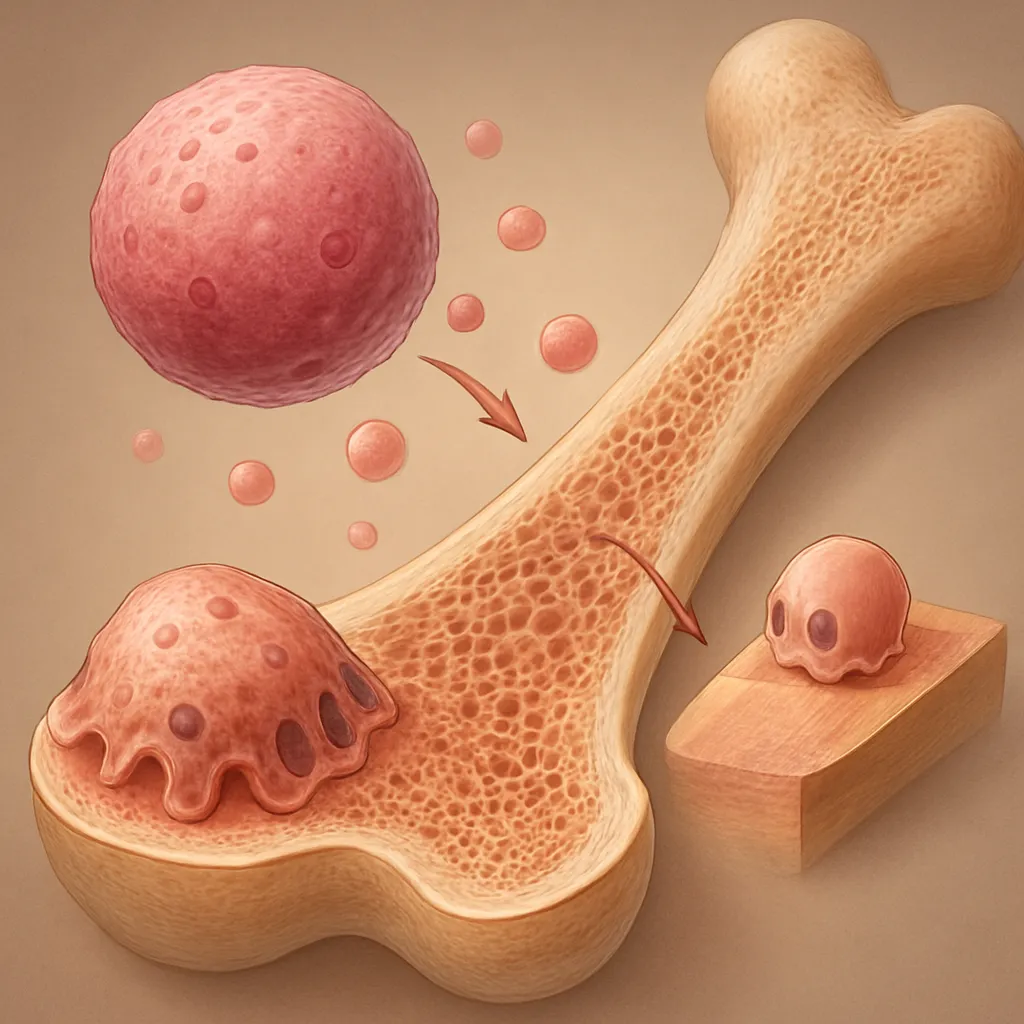
Bone undergoes continuous renewal through a tightly regulated process of osteogenesis and resorption, coordinated by osteoblasts, osteoclasts, and osteocytes. Successful repair of fractures or defects depends on the recruitment of progenitor cells, adequate vascular supply, and balanced signaling from systemic and local mediators. In patients with diabetes, persistent hyperglycemia and the formation of advanced glycation end products (AGEs) compromise matrix quality and cellular viability. Microvascular disease reduces perfusion, while neuropathy limits mechanical loading and proprioception, further hindering appropriate mechanotransduction required for bone healing. In addition, dysregulated production of pro-inflammatory cytokines and oxidative stress interferes with the release of critical growth factors such as bone morphogenetic proteins (BMPs) and vascular endothelial growth factor (VEGF), both essential for effective angiogenesis and new bone formation.
Pathophysiology of Impaired Bone Healing in Diabetic Patients
The disruption of normal healing cascades in diabetes involves multiple interconnected mechanisms:
- Vascular Dysfunction: Endothelial damage and reduced nitric oxide bioavailability lead to microvascular rarefaction, impairing nutrient delivery and removal of waste products during the repair phase.
- Cellular Senescence: High glucose environments induce premature aging of osteoblasts and mesenchymal progenitors, limiting their proliferative and differentiation capacities.
- Altered Immune Response: Chronic inflammation skews macrophage polarization toward a pro-inflammatory M1 phenotype, delaying the transition to the reparative M2 state necessary for matrix deposition.
- Neuropathy: Sensory nerve damage reduces protective reflexes and mechanical stimuli that ordinarily promote callus formation and remodeling.
- Matrix Alterations: Non-enzymatic glycation stiffens collagen fibers, diminishing the biomechanical resilience of newly formed bone and making it prone to microfractures.
- Deficient Stem Cell Niche: Impaired mobilization and homing of mesenchymal stem cells (MSCs) reduce the pool of osteogenic precursors available at injury sites.
Collectively, these factors lead to delayed union, nonunion, and higher rates of infection or implant failure compared to non-diabetic individuals.
Current Therapeutic Approaches
Optimal management of bone defects in diabetic patients necessitates a multifaceted strategy encompassing systemic metabolic control and local regenerative support. Key interventions include:
- Glycemic Optimization: Maintaining hemoglobin A1c within target range reduces AGE accumulation and improves cellular function.
- Pharmacologic Agents: Use of anabolic agents such as teriparatide can stimulate osteoblast activity, while bisphosphonates inhibit excessive resorption. Adjunctive vitamin D or calcium supplementation addresses common deficiencies that compromise mineralization.
- Advanced Biomaterials: Incorporation of bioactive scaffolds loaded with growth factors enhances cellular attachment and guides new tissue formation. Calcium phosphate ceramics and composite polymers have shown promise in preclinical models.
- Autologous and Allogeneic Grafts: Traditional bone grafts remain the gold standard for large defects, but carry risks of donor-site morbidity and limited supply. Innovative synthetic alternatives aim to mimic natural bone matrix while providing controlled degradation.
- Local Drug Delivery: Implantation of carriers releasing anti-inflammatory agents or angiogenic factors directly at the defect site may overcome systemic side effects and concentrate therapeutic molecules where they are most needed.
Despite these options, healing outcomes in diabetic cohorts still lag behind those in healthy populations, prompting exploration of next-generation therapies.
Emerging Strategies and Future Directions
Innovations in regenerative medicine offer new hope for enhancing bone repair under diabetic conditions. Areas of active investigation include:
- Stem Cell Therapies: Transplantation of autologous or allogeneic MSCs, often pre-conditioned in hypoxic environments or genetically modified to overexpress osteogenic genes, can bolster the resident progenitor pool. Combining MSCs with osteoinductive factors such as BMP-2 amplifies their reparative potential.
- Tissue Engineering Platforms: Three-dimensional bioprinting enables fabrication of patient-specific scaffolds incorporating living cells and vascular channels. These constructs aim to recapitulate both the architectural and biological complexity of native bone.
- Nanotechnology: Nanofiber mats and nanoparticle carriers facilitate sustained release of growth factors, peptides, or microRNAs that modulate gene expression in target cells. Surface functionalization with RGD motifs can improve cell adhesion and integration.
- Biomimetic Materials: Development of biomaterials that respond to physiological cues — such as pH-sensitive hydrogels that release drugs upon local acidosis — may enhance targeted therapy in chronically inflamed environments.
- Exosome-Based Therapies: Vesicles derived from stem cells carry proteins and nucleic acids that promote angiogenesis, attenuate apoptosis, and stimulate tissue engineering processes without the risks associated with cell transplantation.
- Gene Editing and Epigenetic Modulation: CRISPR/Cas9 approaches to correct diabetic-induced gene dysregulation in osteoprogenitors hold promise for restoring normal differentiation pathways, while epigenetic drugs may reverse adverse cellular memory induced by hyperglycemia.
Translating these advances from bench to bedside will require rigorous clinical trials and optimized delivery systems. Collaboration among clinicians, material scientists, and bioengineers is essential to overcome the barriers posed by the diabetic milieu and to achieve reliable, long-lasting bone regeneration.