Mechanical loading plays a pivotal role in skeletal health, directly influencing the adaptive growth and strength of bone tissue. Understanding how physical forces translate into cellular responses offers valuable insights into preventing osteoporosis, enhancing rehabilitation protocols, and optimizing athletic performance. This article delves into the intricate processes by which bones sense and respond to mechanical stresses, highlighting key molecular pathways and clinical implications.
Mechanotransduction at the Cellular Level
Mechanosensors in Bone Tissue
Bone cells are equipped with specialized structures that detect and transmit mechanical cues. Among these, the osteocytes—mature bone cells embedded within the mineralized matrix—serve as primary mechanosensors. Their long dendritic processes form an interconnected network, allowing them to perceive local changes in fluid flow and matrix deformation.
Signal Transduction Pathways
Once mechanical forces are detected, a cascade of molecular events unfolds:
- Activation of stretch-sensitive ion channels, allowing calcium influx.
- Engagement of focal adhesion kinases (FAKs), which link integrin receptors to the actin cytoskeleton.
- Initiation of downstream effectors such as mitogen-activated protein kinases (MAPKs).
- Upregulation of anabolic mediators, including prostaglandins and nitric oxide.
- Stimulation of the Wnt signaling pathway, promoting osteogenic gene expression.
Role of the Extracellular Matrix
The extracellular matrix (ECM) provides both structural support and a medium for force transmission. Changes in ECM stiffness modulate the sensitivity of bone cells to mechanical stimuli. Collagen fibers and mineral crystals work synergistically to ensure that strain is distributed efficiently across the tissue, optimizing cellular activation.
Bone Remodeling and Adaptation In Vivo
Patterns of Mechanical Strain
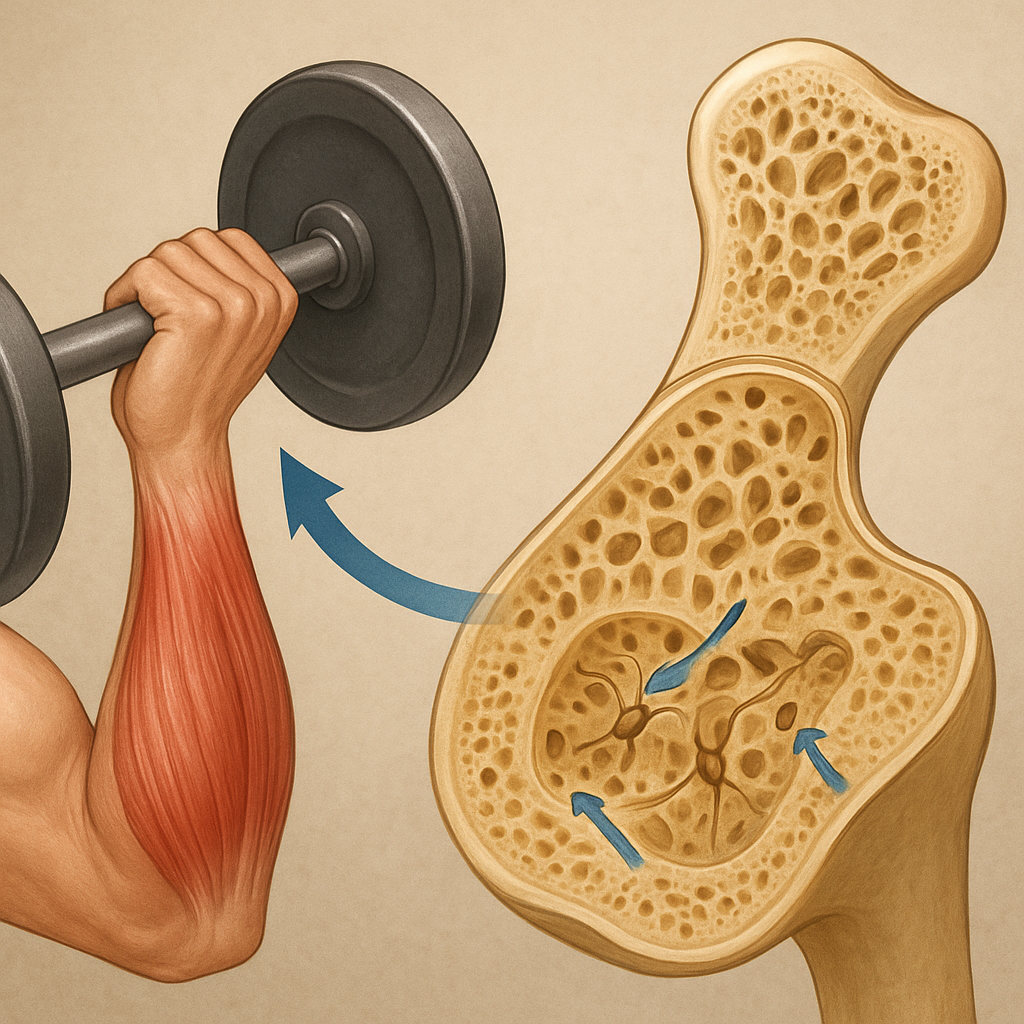
Bone adaptation follows the principles of Wolff’s law: tissues remodel in response to the magnitude, frequency, and duration of applied loads. Key aspects include:
- Strain magnitude: Higher peak strains elicit stronger anabolic responses.
- Loading frequency: Intermittent loading with rest periods is more effective than continuous stress.
- Strain rate: Rapid loading increases fluid shear stress in lacunae, enhancing mechanosensitivity.
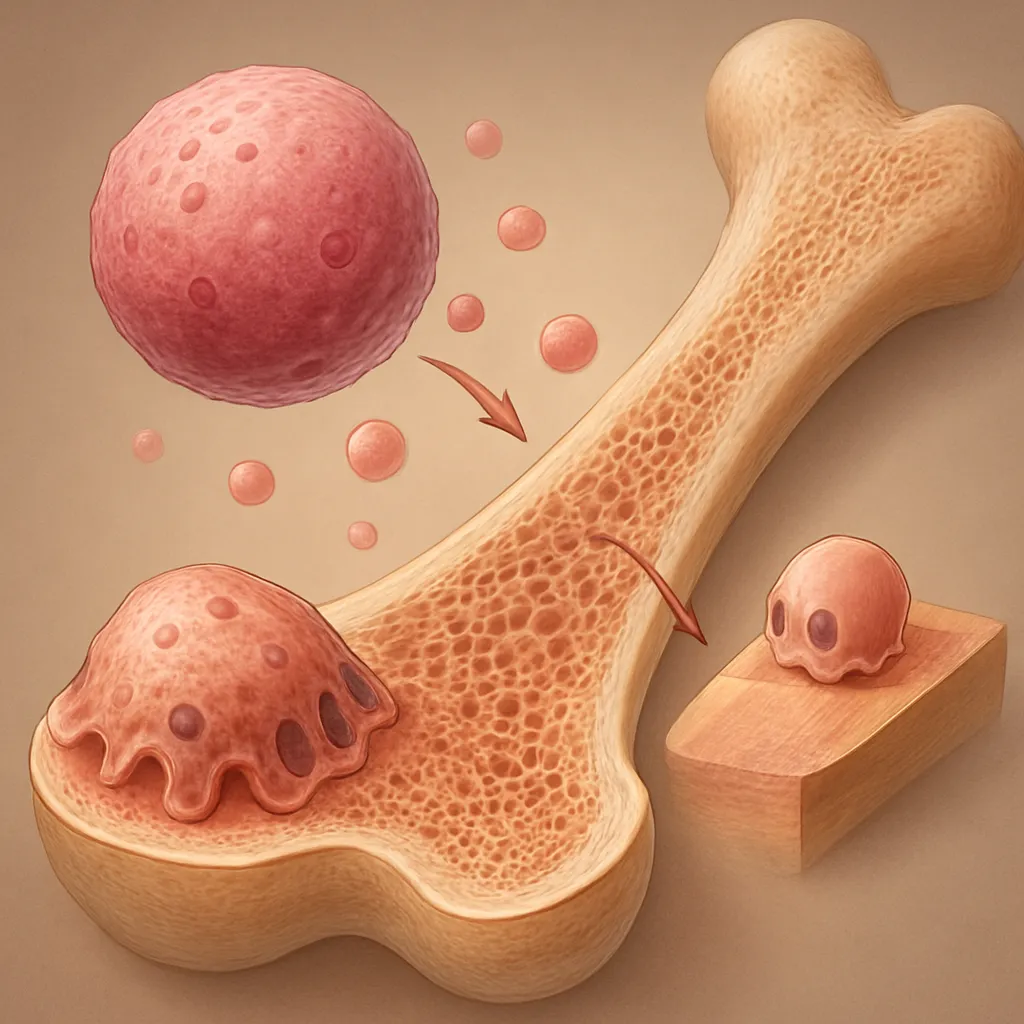
Cellular Contributors to Remodeling
Bone formation and resorption are driven by dynamic interactions among various cell types:
- Osteoblasts: Responsible for new matrix synthesis and mineralization.
- Osteoclasts: Mediators of bone resorption, clearing microdamage and allowing renewal.
- Osteocytes: Regulators of remodeling through secretion of sclerostin, RANKL, and other factors.
Mechanical stimuli shift the balance toward bone formation by downregulating sclerostin (an osteocyte-derived inhibitor) and upregulating anabolic cytokines.
Systemic Influences
Beyond local strain, systemic hormones such as estrogen, parathyroid hormone (PTH), and growth factors modulate the remodeling response. For instance, PTH can enhance the sensitivity of bone cells to mechanical loading, thereby synergizing with exercise interventions.
Clinical and Exercise Applications
Rehabilitation Strategies
Understanding mechanical stimulation informs rehabilitation protocols for patients recovering from fractures or surgery. Controlled weight-bearing exercises and low-magnitude, high-frequency vibration therapy have shown promise in accelerating bone healing by providing mechanical stimuli that activate osteogenic pathways without risking overload.
Osteoporosis Prevention and Treatment
Implementing targeted exercise regimens can increase bone density and reduce fracture risk in at-risk populations. Resistance training, impact loading (e.g., jumping), and balance exercises are recommended to maximize skeletal health. Pharmacological agents that mimic mechanical loading effects are also under development, aiming to potentiate the body’s natural remodeling processes.
Sports Medicine Perspectives
Athletes benefit from optimized loading protocols that enhance bone strength while minimizing injury. Cross-training, periodization of impact activities, and monitoring load thresholds help maintain skeletal integrity. Innovations such as wearable sensors allow real-time assessment of strain patterns, facilitating personalized training plans.
Emerging Directions in Bone Mechanobiology
Biophysical Tools and Imaging
Advanced technologies provide unprecedented insights into mechanotransduction:
- Atomic force microscopy for nanoscale stiffness mapping of bone tissue.
- Micro-CT and high-resolution MRI to visualize microarchitecture changes under load.
- Traction force microscopy to quantify cellular forces in vitro.
Regenerative Medicine and Biomaterials
Engineered scaffolds designed with tunable mechanical properties aim to recreate optimal loading environments for stem cell–mediated bone repair. These biomaterials leverage concepts of remodeling and mechanotransduction to guide tissue regeneration in critical-size defects.
Genetic and Molecular Targeting
Emerging research focuses on modulating key mechanosensitive genes to enhance bone’s adaptive capacity. Strategies include gene editing of sclerostin or integrin subunits and the use of small molecules to amplify signaling pathways such as mechanotransduction.