Osteosarcoma remains one of the most aggressive bone tumors affecting adolescents and young adults. Despite decades of research, the standard combination of surgery and high-dose chemotherapy yields limited improvements in survival once pulmonary metastasis develops. Emerging treatments now focus on exploiting the tumor’s unique biology, the surrounding bone microenvironment, and innovations in drug delivery to overcome therapeutic resistance. Recent preclinical and early clinical studies show promise for combining targeted therapy, immunotherapy, and nanotechnology-based approaches to shift the paradigm of osteosarcoma management.
Background and Tumor Biology
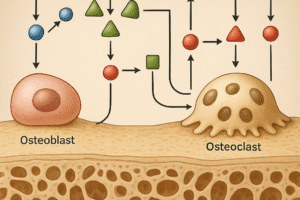
Originally described over a century ago, osteosarcoma is characterized by malignant cells that produce osteoid matrix. Tumor development involves complex interactions between osteoblast precursors, stromal cells, and an extensive vascular network. Critical alterations in molecular pathways such as the p53–Rb axis, Wnt/β-catenin signaling, and PI3K/AKT/mTOR cascade drive unchecked proliferation and survival.
Angiogenic factors like VEGF promote neovascularization to supply nutrients and facilitate metastatic dissemination, particularly to the lungs. The extracellular matrix in bone, rich in hydroxyapatite and collagen, creates a niche that can shelter malignant cells from systemic agents. Additionally, tumor-associated macrophages and myeloid-derived suppressor cells contribute to an immunosuppressive milieu that blunts host anti-tumor responses.
Targeted Molecular Approaches
Efforts to inhibit specific oncogenic drivers have led to the evaluation of small-molecule inhibitors and monoclonal antibodies. By disrupting kinases or receptor–ligand interactions essential for osteosarcoma cell survival, these agents aim to reduce off-target toxicity.
- Tyrosine kinase inhibitors: Agents targeting IGF-1R, PDGFR, and FGFR have shown anti-proliferative effects in preclinical models.
- mTOR inhibitors: Everolimus and temsirolimus block downstream signaling of PI3K/AKT, slowing tumor growth.
- Histone deacetylase inhibitors: Epigenetic modulators that can restore apoptotic pathways in refractory cell lines.
- Angiogenesis blockers: Bevacizumab and multi-kinase inhibitors reduce vascular supply and potentiate chemotherapy.
- Emerging targets: AXL receptor, MET proto-oncogene, and the CDK4/6 axis are under investigation.
Combination strategies pairing targeted agents with conventional chemotherapy may yield synergistic responses by sensitizing tumor cells and disrupting compensatory signaling loops.
Immunotherapy Innovations
Harnessing the immune system offers an alternative route to eliminate minimal residual disease. Vaccines, checkpoint inhibitors, and adoptive cell transfer are at the forefront of this effort:
- Checkpoint blockade: Anti-PD-1/PD-L1 and anti-CTLA-4 antibodies aim to reinvigorate T cell activity against tumor antigens.
- CAR T cell therapy: Chimeric antigen receptor T cells directed at GD2 and HER2 have been engineered to recognize osteosarcoma cells.
- Oncolytic viruses: Genetically modified viruses selectively replicate in cancer cells, inducing immunogenic cell death.
- Peptide and dendritic cell vaccines: Tumor-associated antigens are presented to prime cytotoxic lymphocytes.
While monotherapy has yielded modest results, combination with anti-angiogenic agents or nanotechnology-driven delivery of immunomodulators may overcome the immunosuppressive tumor microenvironment. Biomarker-driven patient selection is essential to identify individuals most likely to benefit from specific immunotherapeutic regimens.
Nanotechnology and Gene-Based Strategies
Advanced delivery platforms can enhance drug accumulation within the tumor bed and minimize systemic toxicity. Nanoparticles, liposomes, and polymeric micelles have been engineered to carry cytotoxic drugs, siRNA, or CRISPR/Cas9 components directly to malignant osteoblastic cells.
Nanocarrier Advantages
- Enhanced permeability and retention (EPR) effect facilitates passive targeting of tumor tissue.
- Surface modification with antibodies or peptides enables active targeting of osteosarcoma-specific antigens.
- Controlled release kinetics prolongs drug exposure and overcomes rapid clearance.
Gene Editing and RNA Interference
- CRISPR/Cas9: Enables knockout of driver oncogenes or insertion of suicide genes into tumor cells.
- siRNA and shRNA nanoparticles: Silence expression of Bcl-2, MDM2, or other survival factors to induce apoptosis.
- MicroRNA mimics: Restore tumor-suppressive miRNAs lost in osteosarcoma.
These approaches are still in early-phase trials but demonstrate the potential to precisely correct malignant phenotypes, offering hope for refractory disease resistant to chemotherapy and radiation.
Precision Medicine and Biomarker Development
Moving beyond one-size-fits-all treatment, precision oncology leverages biomarkers and genomic profiling to tailor therapy. High-throughput sequencing of tumor samples identifies actionable mutations, copy number variations, and gene fusions. Liquid biopsies detecting circulating tumor DNA (ctDNA) may provide real-time monitoring of disease burden and emerging resistance.
Key areas of research include:
- Integration of multi-omic data (genomic, transcriptomic, proteomic) to stratify patients.
- Development of predictive assays for drug sensitivity, such as organoid cultures derived from patient biopsies.
- Exploration of metabolic biomarkers that reflect alterations in glycolysis, glutaminolysis, and lipid metabolism specific to osteosarcoma cells.
By correlating biomarker profiles with clinical outcomes, researchers aim to optimize combination regimens, minimize unnecessary toxicity, and improve long-term survival in osteosarcoma patients.