The integrity of the skeletal system depends not only on osteoblasts and osteoclasts but also on the often-overlooked osteocytes. Embedded deep within the mineralized matrix, these cells orchestrate a complex interplay of signals that ensure bone strength and adaptability. Understanding their multifaceted roles is crucial for advancing treatments for osteoporosis, fractures, and other metabolic bone diseases.
Osteocyte Biology and Microenvironment
Osteocytes originate from mature osteoblasts that become entrapped in the extracellular matrix during bone formation. Once embedded, they establish an extensive network of dendritic processes that traverse tiny channels called canaliculi. This lacuno-canalicular system allows osteocytes to communicate with surface cells and sense mechanical and biochemical cues.
Cell Differentiation and Morphology
As osteoblasts differentiate into osteocytes, they undergo remarkable morphological changes. Their cell bodies shrink, and they extend long cytoplasmic processes. These processes connect with neighboring osteocytes via gap junctions, mainly composed of connexin proteins. Such connectivity underpins the rapid transmission of signals throughout the bone matrix.
Matrix Mineralization
Osteocytes control local mineralization by regulating the deposition and removal of hydroxyapatite crystals. They secrete proteins such as matrix extracellular phosphoglycoprotein (MEPE) and dentin matrix protein 1 (DMP1), which modulate crystal growth. By fine-tuning the mineral content, osteocytes optimize bone stiffness and resilience.
Mechanotransduction and Signaling Pathways
Mechanical loading stimulates osteocytes to convert physical forces into biochemical responses—a process known as mechanotransduction. This capacity enables the skeleton to adapt to varying mechanical demands, preventing bone loss in low-load conditions and promoting remodeling in high-load scenarios.
Fluid Shear Stress and Ion Channels
During mechanical loading, interstitial fluid moves through the canaliculi, generating shear stress on osteocyte processes. Ion channels, including TRPV4 and PIEZO1, detect these forces, causing intracellular calcium influx. Elevated calcium triggers downstream cascades that influence gene expression and protein secretion.
Sclerostin: A Central Inhibitor
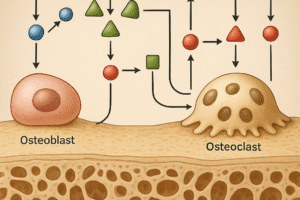
One of the most critical osteocyte-derived regulators is sclerostin, a glycoprotein encoded by the SOST gene. Sclerostin binds to LRP5/6 receptors on osteoblasts and pre-osteoblasts, antagonizing the Wnt/β-catenin pathway. Under mechanical load, osteocytes reduce sclerostin production, thereby enhancing Wnt signaling and promoting bone formation. Conversely, increased sclerostin during disuse leads to bone resorption.
Other Signaling Molecules
- RANKL (Receptor Activator of Nuclear Factor κB Ligand): Osteocyte-derived RANKL stimulates osteoclast differentiation, balancing formation and resorption.
- FGF23: Regulates systemic phosphate homeostasis by acting on kidney transporters.
- Prostaglandin E2 (PGE2): Produced in response to mechanical stimuli, modulates osteoblast activity.
Clinical Implications and Therapeutic Opportunities
Insights into osteocyte function have fueled novel approaches for treating metabolic bone disorders. Targeting osteocyte-specific pathways offers the opportunity to enhance bone mass and quality without the side effects associated with traditional therapies.
Anti-Sclerostin Antibodies
Monoclonal antibodies against sclerostin, such as romosozumab, have demonstrated significant increases in bone mineral density and reduced fracture risk in clinical trials. By neutralizing sclerostin, these agents lift the inhibitory brake on Wnt signaling, stimulating osteoblast proliferation and activity.
Modulating RANKL and OPG
Balancing RANKL and osteoprotegerin (OPG) levels is another strategy. Denosumab, a monoclonal antibody against RANKL, prevents osteoclast maturation, thereby reducing bone resorption. Understanding osteocyte-derived RANKL sources can refine dosing regimens to maximize efficacy and safety.
Mechanotherapy and Biophysical Approaches
Physical therapies that apply controlled mechanical stimuli, such as vibration platforms or low-intensity pulsed ultrasound, aim to harness mechanotransduction pathways. By stimulating osteocytes directly, these interventions may promote bone formation and mitigate disuse osteoporosis, especially in immobilized patients or astronauts exposed to microgravity.
Emerging Molecular Targets
- Connexin43 Channels: Modulators that enhance gap-junction communication could improve signal propagation and bone adaptation.
- PIEZO1 Agonists: Agents that sensitize mechanosensitive channels may amplify bone’s response to mechanical load.
- Epigenetic Regulators: Targeting microRNAs or histone modifiers that control osteocyte gene expression opens avenues for fine-tuned interventions.
Future Directions in Osteocyte Research
Advancements in imaging, single-cell transcriptomics, and 3D culture systems are unraveling osteocyte heterogeneity and niche-specific functions. Profiling osteocytes at various skeletal sites will illuminate how microenvironmental differences affect their roles in health and disease. Ultimately, integrating these findings with systems biology models promises to revolutionize our ability to predict fracture risk and personalize treatments.