The growing interest in exercise protocols that not only improve cardiovascular fitness but also enhance skeletal health has led researchers to investigate how high-intensity training impacts bone structure. This article explores the physiological mechanisms, experimental findings, and practical applications of high-intensity exercise for optimizing skeletal integrity.
Physiological Mechanisms Underlying Bone Adaptation
Mechanotransduction Pathways
When mechanical loads are applied to bone, specialized cells convert physical forces into biochemical signals. These signals trigger a cascade of events via integrins and cytoskeletal networks, ultimately leading to increased osteogenesis. The strain magnitude, frequency, and rate are critical variables: rapid, high-magnitude loads activate signaling through the Wnt/β-catenin pathway and upregulate nitric oxide synthase, enhancing cellular proliferation.
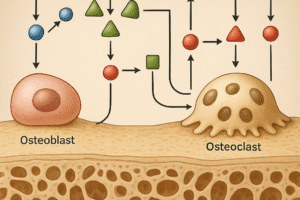
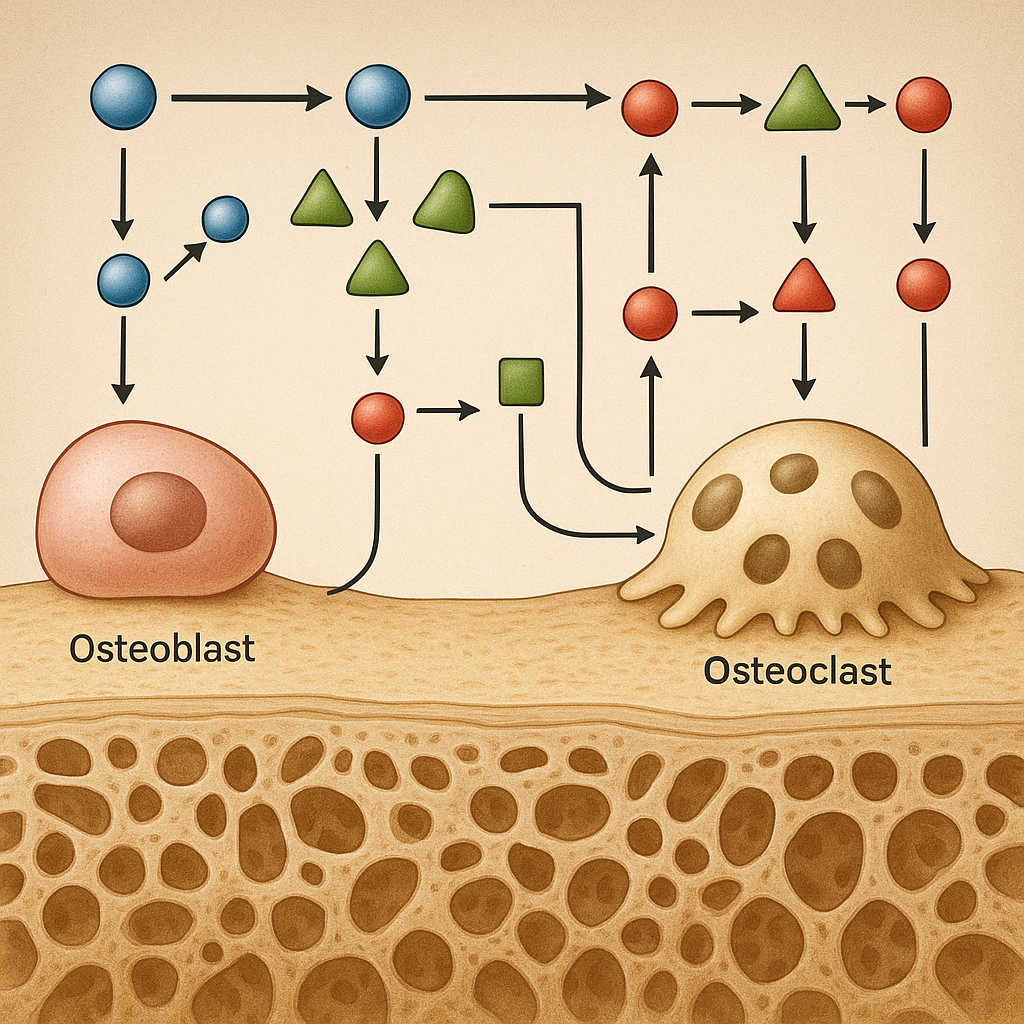
Cellular Responses: Osteoblasts vs Osteoclasts
Skeletal remodeling involves a tight balance between bone-forming osteoblasts and bone-resorbing osteoclasts. High-intensity loading stimulates osteoblast differentiation via elevated expression of Runt-related transcription factor 2 (Runx2). Concurrently, mechanical stress alters the RANK/RANKL/OPG axis, reducing osteoclast-mediated resorption. This shift increases the ratio of bone formation to resorption, promoting net accrual of bone mass.
Experimental and Clinical Evidence
Insights from Animal Models
Rodent models subjected to high-frequency vibration or treadmill running at steep inclines demonstrate marked improvements in both cortical and trabecular bone microarchitecture. Micro-CT analyses reveal increased trabecular thickness, reduced separation, and a more connected network. Biomechanical testing highlights improved stiffness and maximum load capacity. These findings underscore the importance of load direction and dynamic oscillations in modeling bone’s adaptive response.
Human Intervention Trials
Clinical trials involving athletes and sedentary individuals compare high-intensity interval training (HIIT) with moderate continuous exercise. Dual-energy X-ray absorptiometry (DXA) and peripheral quantitative CT (pQCT) assessments confirm that short bouts of vigorous loading yield significant gains in bone mineral density at the lumbar spine and femoral neck. Blood biomarkers such as bone-specific alkaline phosphatase and C-terminal telopeptide reflect heightened turnover rates during early adaptation phases.
Impact on Bone Geometry and Microarchitecture
Cortical Bone Modifications
Under high-strain conditions, periosteal apposition increases, causing a modest expansion of bone diameter. This change enhances the section modulus, improving resistance to bending and torsional forces. Finite element models demonstrate that even a 5% increase in cortical thickness can lead to a 15% rise in overall bone strength.
Trabecular Network Enhancements
High-intensity loads preferentially target trabecular compartments due to the transfer of dynamic compressive forces. Increased connectivity density and reduced trabecular spacing augment load distribution. These microarchitectural improvements mitigate stress concentrations that can precipitate microfractures under repetitive loading.
Implications for Clinical Practice and Rehabilitation
Integrating high-intensity protocols into therapeutic regimens can benefit populations at risk of osteoporosis and stress fractures. Key considerations include individual risk assessment, exercise progression, and monitoring to avoid overtraining injuries. The following principles guide safe implementation:
- Individualization: Tailor program intensity based on baseline bone health and functional capacity.
- Progressive Overload: Gradually increase load magnitude and frequency to stimulate continued adaptation.
- Recovery: Allow sufficient rest between sessions to permit cellular repair and minimize cumulative microdamage.
- Cross-Training: Incorporate varied loading modalities such as plyometrics, resistance exercises, and agility drills.
- Monitoring: Use imaging and biochemical markers to track skeletal responses and adjust the regimen accordingly.
Future Directions and Research Opportunities
Despite promising results, several questions remain unanswered. Long-term safety of sustained high-intensity loading requires further investigation, as does the optimal dose–response relationship for different age groups and genders. Advances in wearable technology and real-time biomechanical feedback hold potential for precision exercise prescription. Moreover, elucidating molecular mechanisms at the epigenetic level may open avenues for pharmacological augmentation of exercise-induced bone gains.
By bridging the gap between exercise science and bone biology, high-intensity training protocols can be refined to maximize skeletal health outcomes. Continued collaboration among clinicians, biomechanists, and molecular biologists will be essential to translate these insights into practical guidelines that enhance bone strength while minimizing injury risk.