The interplay between Vitamin C and bone collagen represents a critical nexus in skeletal physiology. This vitamin acts as a vital cofactor in the synthesis and stabilization of collagen, which constitutes the primary organic framework of bone tissue. Deficiencies can weaken the extracellular matrix, impair osteogenic processes, and predispose individuals to structural fragility. Understanding the molecular and clinical aspects of this relationship sheds light on potential therapeutic interventions aimed at preserving bone density and reducing the incidence of fracture-related morbidity.
Understanding the Role of Vitamin C in Collagen Biosynthesis
Biochemical Pathways
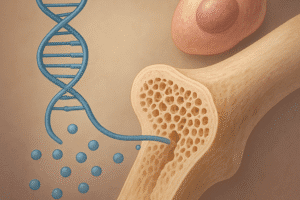
At the heart of collagen production lies a complex enzymatic cascade. The post-translational modifications essential for mature collagen formation include proline and lysine hydroxylation. These reactions are catalyzed by prolyl and lysyl hydroxylase enzymes, both requiring Vitamin C as an indispensable reducing agent. Without adequate vitamin levels, reduced enzyme activity leads to under-hydroxylated collagen chains that cannot form stable triple helices, compromising tensile strength and resilience.
Enzymatic Hydroxylation and Collagen Stability
Following translation of procollagen into the rough endoplasmic reticulum, prolyl and lysyl hydroxylases introduce hydroxyl groups at specific residues. This modification allows glycosylation and the eventual assembly of three alpha chains. The stabilization of the triple-helix conformation hinges on these hydroxyl groups forming interchain hydrogen bonds. A deficiency in hydroxyproline, the hydroxylated proline derivative, disrupts helix formation, yielding poorly cross-linked fibrils and diminishing overall collagen stability.
Impact on Bone Health and Density
Osteoblast Function and Matrix Formation
Osteoblasts secrete osteoid, the unmineralized organic component of bone, primarily composed of collagen type I. Optimal osteoid synthesis depends on the preservation of hydroxylase activity. Vitamin C fosters osteogenic differentiation by upregulating genes such as RUNX2 and enhancing alkaline phosphatase activity. Moreover, it exerts immunomodulatory effects by modulating cytokine production, indirectly promoting a favorable environment for bone deposition.
Animal and Clinical Studies
Preclinical trials in ascorbate-deficient animal models demonstrate reduced bone formation rates, decreased bone volume, and a higher prevalence of microfractures. Clinical observational studies correlate low plasma ascorbate levels with diminished bone mineral density in postmenopausal women and elderly men. Interventional trials supplementing vitamin C show modest improvements in bone turnover markers, suggesting potential benefits in high-risk populations, although long-term fracture data remain limited.
Vitamin C Deficiency and Skeletal Disorders
Scurvy and Bone Fragility
Scurvy, the classical disease of severe Vitamin C deficiency, manifests with gum bleeding, poor wound healing, and musculoskeletal symptoms. In children, it can present as pseudoparalysis due to subperiosteal hemorrhages and defective osteoid formation. Radiographic signs include scorbutic rosary and cortical thinning. Adults may experience bone pain, swelling, and spontaneous fractures, reflecting compromised collagen integrity throughout the skeletal system.
Subclinical Deficiency and Long-term Risks
Even marginal insufficiency can impair collagen cross-linking, leading to microarchitectural deterioration over decades. Epidemiological data link low daily intake to increased incidence of osteoporosis, particularly when combined with insufficient dietary calcium and vitamin D. Oxidative stress exacerbates collagen degradation, further undermining bone strength. Recognition of subclinical deficiency is critical, as early intervention could forestall progressive skeletal decline.
Therapeutic Implications
Dietary Intake and Supplementation
Dietary recommendations for adults typically range from 75 to 90 mg per day, yet many individuals fail to meet this threshold through fruit and vegetable consumption alone. Foods richest in Vitamin C include citrus fruits, bell peppers, and cruciferous vegetables. Supplementation strategies vary from low-dose daily formulations to high-dose regimens in clinical settings. Bioavailability studies indicate diminishing returns above 200 mg per dose, urging fractionated dosing throughout the day for optimal plasma saturation.
Potential in Osteoporosis Management
Emerging evidence suggests that adjunctive vitamin C supplementation may complement standard antiresorptive and anabolic therapies. In vitro studies reveal that ascorbate enhances the efficacy of parathyroid hormone analogs by promoting matrix maturation. Additionally, its antioxidant properties may protect osteoblasts from oxidative insults linked to estrogen deficiency. However, large randomized trials are needed to confirm fracture reduction benefits and establish precise dosing protocols.
Future Directions in Research
Novel Biomarkers
Advancements in omics technologies offer opportunities to identify sensitive indicators of collagen turnover. Measuring circulating propeptides of type I procollagen and advanced glycation end products may provide real-time insights into matrix dynamics in response to vitamin C intake. Integration of metabolomic profiles could further elucidate the interplay between micronutrient status and bone remodeling pathways.
Clinical Trial Designs
Future studies should incorporate robust fracture endpoints and stratify participants by baseline nutritional status and genetic polymorphisms affecting ascorbate transport. Multi-arm trials comparing monotherapy with combination approaches involving vitamin D, calcium, and vitamin C will clarify synergistic effects. Additionally, exploring the role of localized high-dose ascorbate delivery via biomaterial scaffolds could revolutionize regenerative strategies for bone defects.