The process of bone remodeling is an intricate ballet of cellular interactions and molecular signals orchestrating the renewal and repair of skeletal tissue. Maintaining a dynamic balance between bone formation and resorption is crucial not only for structural integrity but also for metabolic homeostasis. This article delves into the fundamental mechanisms that govern bone remodeling, explores the most critical signaling networks, and examines their translational significance in modern medicine.
Molecular Framework of Bone Homeostasis
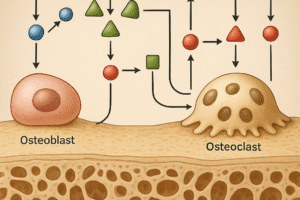
Bone is a living tissue that adapts continuously to mechanical forces and chemical cues. Two primary cell populations drive this adaptation: osteoblasts, which lay down new bone matrix, and osteoclasts, specialized in resorption. The synchronized activity of these cells forms the basis of the remodeling cycle, consisting of activation, resorption, reversal, formation, and termination phases.
Osteoclast Differentiation and Function
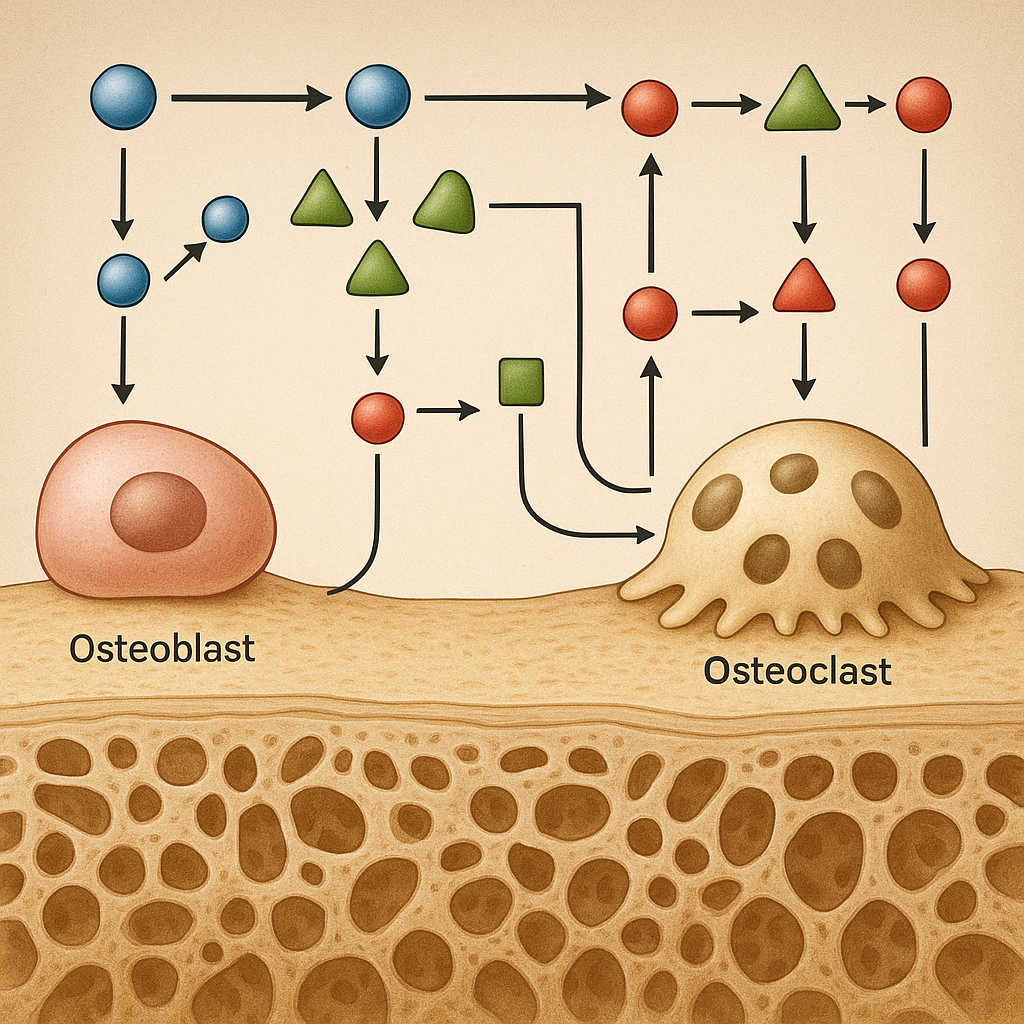
Originating from hematopoietic precursors, osteoclasts undergo terminal differentiation in response to local factors. A key regulatory axis is the RANK/RANKL system: receptor activator of nuclear factor κB (RANK) on osteoclast precursors binds its ligand (RANKL) expressed by osteoblast-lineage cells. This interaction triggers recruitment of adaptor proteins, activation of NF-κB, and upregulation of genes essential for multinucleated cell fusion and acidification machinery.
Osteoblast Lineage Commitment
Mesenchymal stem cells differentiate into osteoblasts under the influence of growth factors such as bone morphogenetic proteins. BMPs activate SMAD transcription factors, promoting expression of RUNX2 and osterix, master regulators of osteoblastogenesis. Mature osteoblasts secrete type I collagen and non-collagenous proteins, establishing a scaffold for mineral deposition.
Coupling Signals
Coupling between formation and resorption involves multiple mediators. Osteoclasts release matrix-embedded growth factors during resorption, including transforming growth factor β (TGF-β) and insulin-like growth factors (IGFs), which recruit osteoblast progenitors. Conversely, osteoblasts secrete osteoprotegerin (OPG), a decoy receptor that binds RANKL to temper osteoclastogenesis, ensuring balanced remodeling.
Principal Signaling Pathways
Complex signaling networks fine-tune bone cell activity in response to systemic hormones, local cytokines, and mechanical stimuli. Below are the pivotal pathways governing skeletal homeostasis.
Wnt/β-Catenin Pathway
The Wnt family of glycoproteins is essential for osteoblast proliferation and survival. Binding of Wnt ligands to the Frizzled/LRP5-6 receptor complex inhibits GSK-3β, stabilizing β-catenin. Nuclear β-catenin then drives transcription of osteogenic genes. Dysregulation of Wnt signaling contributes to osteoporosis and skeletal anomalies.
RANKL-Mediated NF-κB and MAPK Cascades
When RANKL engages RANK on osteoclast precursors, it recruits TRAF6, leading to activation of NF-κB and MAPK pathways. NF-κB translocates to the nucleus, upregulating critical osteoclastogenic transcription factors like NFATc1. Simultaneously, MAPKs (ERK, p38, JNK) modulate cell survival, differentiation, and cytoskeletal organization essential for resorptive function.
PI3K/Akt and mTOR Signaling
The PI3K/Akt axis supports both osteoblast and osteoclast survival. Activation of PI3K generates PIP3, recruiting Akt to the plasma membrane where it becomes phosphorylated. Akt phosphorylates downstream targets, including mTOR, facilitating protein synthesis and cytoskeletal remodeling. This pathway intersects with Wnt and growth factor signaling to coordinate cellular metabolism with bone turnover.
Cytokines and Hormonal Modulation
Inflammatory cytokines such as interleukin-1 (IL-1), interleukin-6 (IL-6), and tumor necrosis factor α (TNFα) enhance osteoclastogenesis by upregulating RANKL and downregulating OPG. Parathyroid hormone (PTH) exerts dual effects: intermittent PTH treatment stimulates bone formation, while continuous exposure triggers resorption. Estrogen maintains skeletal mass by inhibiting osteoclast activity and fostering OPG expression.
Clinical Applications and Novel Therapies
Understanding bone signaling has revolutionized therapeutic strategies for osteoporosis, fracture healing, and metabolic bone disorders. Several interventions now target specific molecular pathways to restore skeletal balance.
Anti-RANKL Antibodies
Denosumab, a human monoclonal antibody against RANKL, effectively suppresses osteoclast formation and activity. Clinical trials demonstrate significant increases in bone mineral density and reduction in fracture risk among postmenopausal women. Its mechanism directly exploits knowledge of the RANK/RANKL pathway to curb pathological resorption.
Wnt Pathway Modulators
Romosozumab, an antibody against sclerostin, enhances Wnt signaling by preventing inhibition of LRP5/6 co-receptors. Treatment leads to robust bone formation and decreased resorption. By manipulating the Wnt network, this agent offers a dual anabolic and antiresorptive effect, representing a major advance in osteoporosis management.
BMP-Based Approaches
Recombinant BMPs have been applied in spinal fusion and fracture nonunion to stimulate local bone formation. Delivery systems such as collagen sponges and hydrogel carriers optimize BMP bioactivity. While effective, meticulous dosage control is critical to minimize adverse effects like ectopic ossification.
Emerging Targets and Future Directions
- Inhibitors of cathepsin K, an osteoclast-derived protease essential for collagen degradation.
- Small molecules modulating PI3K/Akt signaling to fine-tune cell survival and anabolic responses.
- Gene therapies delivering RUNX2 or OPG constructs to enhance osteoblastogenesis and suppress resorption.
- Biomaterial scaffolds incorporating mechanical loading paradigms to harness mechanotransduction for improved bone regeneration.
Ongoing research into the intricate crosstalk among growth factors, hormones, and mechanical forces continues to expand our arsenal against skeletal diseases. By integrating molecular insights with clinical innovation, the field moves closer to fully personalized strategies for maintaining and restoring bone health.