The intricate relationship between human immunodeficiency virus (HIV) and skeletal physiology has emerged as a critical concern in modern medicine. Growing evidence indicates that both the virus itself and its therapeutic interventions can disrupt **bone mineral density**, leading to heightened fracture risk. Understanding the multifaceted mechanisms underlying bone loss in people living with HIV is essential for optimizing long-term care and improving quality of life.
Impact of HIV Infection on Bone Remodeling
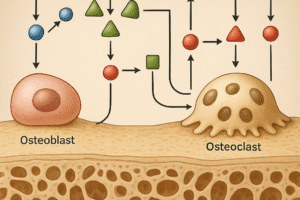
HIV infection drives a persistent state of **inflammation** and **immune activation** that profoundly alters the delicate equilibrium of bone turnover. Under normal conditions, bone homeostasis relies on a synchronized interplay between **osteoclasts** (bone-resorbing cells) and **osteoblasts** (bone-forming cells). In individuals with untreated HIV, several factors shift this balance toward net bone loss:
- Pro-inflammatory Cytokines: Elevated levels of tumor necrosis factor-alpha (TNF-α), interleukin-6 (IL-6), and interleukin-1β (IL-1β) enhance osteoclastogenesis by upregulating RANKL expression on osteoblasts and stromal cells.
- RANKL/OPG Imbalance: The ratio of receptor activator of nuclear factor κB ligand (RANKL) to osteoprotegerin (OPG) is skewed, favoring osteoclast differentiation and activation.
- Direct Viral Effects: HIV proteins such as gp120 and Vpr can induce osteoblast apoptosis, impairing bone formation capacity.
- Immune Cell Dysregulation: Chronic T-cell activation alters bone marrow microenvironment, reducing osteoprogenitor cell proliferation.
These disturbances manifest in reduced **bone mineral density** (BMD) detectable on DEXA scans as early as the first year of infection. Without timely intervention, this accelerated bone loss heightens the risk of osteoporosis and fragility fractures in a relatively young population.
Effects of Antiretroviral Therapy on Skeletal Health
While antiretroviral therapy (ART) has transformed HIV into a chronic manageable condition, certain regimens exert unintended skeletal side effects:
Nucleoside and Nucleotide Reverse Transcriptase Inhibitors (NRTIs)
- Tenofovir Disoproxil Fumarate: Associated with proximal renal tubulopathy and phosphate wasting, resulting in decreased BMD.
- Stavudine and Zidovudine: Linked to mitochondrial toxicity and lactic acidosis, indirectly affecting bone health through muscle weakness and reduced mobility.
Protease Inhibitors (PIs)
- Some PIs may impair vitamin D metabolism by inhibiting 1α-hydroxylase in the kidney.
- Alterations in lipid profiles and fat redistribution can exacerbate inflammation and oxidative stress.
Integrase Strand Transfer Inhibitors (INSTIs) and Non-Nucleoside Reverse Transcriptase Inhibitors (NNRTIs)
- Generally considered to have a more favorable bone safety profile, though long-term data are still emerging.
The initiation of ART often triggers an initial 2–6% decrease in BMD within the first 48–96 weeks, a phenomenon termed the “ART-associated bone loss.” Effective viral suppression later stabilizes bone turnover but seldom reverses the early deficit. Balancing viral control against skeletal risks is a major clinical challenge.
Clinical Consequences and Management Strategies
Given the elevated fracture risk among people with HIV, comprehensive assessment and proactive management are imperative. Key strategies include:
- Screening and Monitoring:
- Annual DEXA scans for patients over 50 or those with additional risk factors.
- Regular evaluation of serum calcium, phosphate, and 25-hydroxyvitamin D levels.
- Lifestyle Modifications:
- Weight-bearing exercises to stimulate osteogenesis.
- Smoking cessation and moderation of alcohol intake.
- Diet rich in calcium and protein.
- Supplementation:
- Vitamin D (cholecalciferol) to achieve serum levels above 30 ng/mL.
- Calcium intake of 1,000–1,200 mg daily through diet or supplements.
- Pharmacotherapies:
- Bisphosphonates (e.g., alendronate) to inhibit osteoclast-mediated resorption.
- Denosumab, a RANKL inhibitor, for patients intolerant to bisphosphonates.
- Selective estrogen receptor modulators (SERMs) in postmenopausal women.
- ART Modification: Considering tenofovir alafenamide (TAF) in place of tenofovir DF or selecting INSTI-based regimens when clinically appropriate.
Early intervention can mitigate bone loss and reduce fracture incidence. Interdisciplinary collaboration among infectious disease specialists, endocrinologists, and orthopedic surgeons ensures holistic patient care.
Emerging Research and Future Directions
Ongoing investigations aim to unravel novel therapeutic targets and refine preventive strategies:
- Modulation of Wnt Signaling: Enhancing osteoblast differentiation by inhibiting antagonists such as sclerostin.
- Advanced Biomarkers: Utilizing bone turnover markers (e.g., P1NP, CTX) for early detection of imbalances before BMD loss becomes apparent.
- Microbiome–Bone Axis: Exploring how gut dysbiosis in HIV influences nutrient absorption and systemic inflammation.
- Gene Therapy: Investigating gene-editing approaches to correct immune dysregulation and protect skeletal integrity.
As life expectancy in HIV-positive populations approaches that of the general population, safeguarding skeletal health will remain a cornerstone of long-term management. Continued research holds promise for interventions that not only suppress viral replication but also preserve and restore bone strength.