Bone health relies on a delicate balance between formation and breakdown, with **resorption** playing a pivotal role in maintaining skeletal integrity. Disruption of this equilibrium can lead to debilitating conditions, increasing the risk of fractures and impairing quality of life. This article explores the mechanisms of bone resorption, highlights major diseases driven by excessive breakdown, and examines cutting-edge **therapeutic** strategies designed to restore bone strength.
Understanding the Biology of Bone Resorption
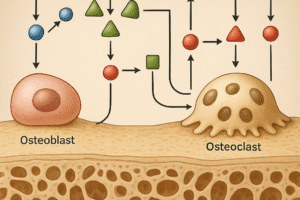
Bone is a dynamic tissue constantly remodeled by two cellular players: osteoclasts, responsible for **resorption**, and osteoblasts, which drive new bone **formation**. The process is regulated by a network of signaling molecules, including the RANK/RANKL/OPG axis. RANKL (Receptor Activator of Nuclear Factor κB Ligand) binds to RANK on osteoclast precursors, stimulating their differentiation and activation. Osteoprotegerin (OPG) acts as a decoy receptor for RANKL, inhibiting excessive osteoclast activity.
Key Molecular Mediators
- RANKL: Master regulator of osteoclast development.
- OPG: Protective factor that prevents overactive resorption.
- Calcitonin: Hormone that directly inhibits osteoclasts.
- Parathyroid hormone (PTH): Dual role; intermittent exposure is anabolic, chronic exposure is catabolic.
Major Diseases Characterized by Excessive Bone Resorption
When resorption outpaces formation, bone becomes porous and fragile. Several disorders manifest through this imbalance:
Osteoporosis
- Most prevalent metabolic bone disease.
- Characterized by reduced bone mass and microarchitectural deterioration.
- High risk of vertebral, hip, and wrist **fractures**.
Paget’s Disease of Bone
- Localized areas of accelerated remodeling.
- Bone enlargement leads to deformities and pain.
- Overactive osteoclasts followed by chaotic osteoblast recruitment.
Bone Metastases and Lytic Lesions
- Cancers such as breast and prostate can metastasize to bone.
- Tumor cells secrete PTH-related protein, stimulating osteoclasts.
- Leads to severe pain and pathological fractures.
Modern Antiresorptive Therapies
Treatments aim to curb osteoclast activity, thereby preserving bone density and reducing fracture risk. Key agents include:
Bisphosphonates
- Mechanism: Adsorb to bone mineral and induce osteoclast apoptosis.
- Examples: Alendronate, risedronate, zoledronic acid.
- Advantages: Oral and intravenous formulations; proven efficacy in fracture prevention.
- Adverse effects: Gastrointestinal irritation, osteonecrosis of the jaw (rare).
Denosumab
- Mechanism: Monoclonal antibody targeting **RANKL**, preventing osteoclast formation.
- Administered subcutaneously every six months.
- Reduces vertebral and nonvertebral fracture risk.
- Potential rebound bone loss upon discontinuation; careful monitoring required.
Sclerostin Inhibitors
- Sclerostin, produced by osteocytes, inhibits bone formation.
- Antibodies (e.g., romosozumab) block sclerostin, increasing bone mass through combined anabolic and antiresorptive effects.
- Effective in severe osteoporosis; cardiovascular safety under review.
Emerging and Adjunctive Treatment Strategies
Research continues to unveil novel targets and combination approaches to optimize bone health:
Cathepsin K Inhibitors
- Cathepsin K is a protease vital for collagen degradation by osteoclasts.
- Selective inhibitors (e.g., odanacatib) have shown promise but face safety concerns.
Anabolic Agents
- Teriparatide: Recombinant PTH fragment; stimulates osteoblast activity when given intermittently.
- Useful for patients with very **low** bone density or multiple fractures.
- Limited to two years of use due to theoretical risk of osteosarcoma.
Combination and Sequential Therapies
Sequential use of antiresorptives and anabolic agents can maximize gains in bone density. For instance, initiating treatment with teriparatide and following with denosumab may yield superior results compared to monotherapy. Personalized regimens based on patient risk factors and treatment response are increasingly common.
Challenges and Future Directions
Despite advances, several hurdles remain in managing resorptive bone diseases. Identification of biomarkers to predict fracture risk and treatment response is a key priority. Additionally, long-term safety data for newer agents will shape clinical guidelines. Ongoing **clinical** trials are exploring gene therapies, gut microbiome modulation, and targeted nanomedicine to further refine interventions.