Bone tissue endures a lifetime of mechanical stresses that can produce microscopic damage before any overt fracture occurs. Subtle fissures, referred to as bone microcracks, accumulate in the cortex and trabeculae, triggering a finely tuned cascade of cellular events. Understanding how the skeletal system detects and repairs such microdamage is crucial for advancing treatments for osteoporosis, stress fractures and other skeletal disorders.
Bone Microcrack Formation and Detection
Mechanisms of Microcrack Generation
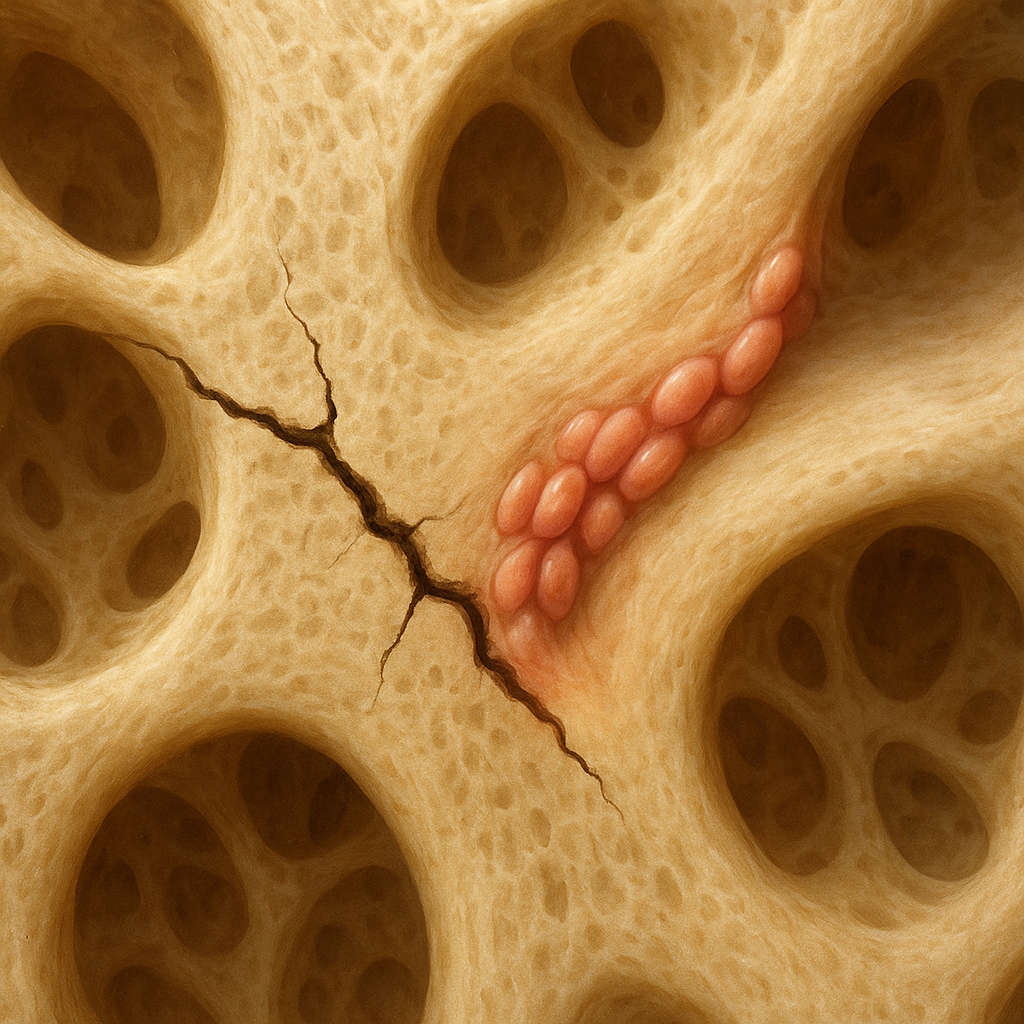
Repeated cyclic loading during routine activities or athletic training induces fatigue in the mineralized matrix of bone. Under tensile and compressive forces, mineral crystallites and the organic collagen network develop small separations. When local stress exceeds the yield threshold, these separations coalesce into microscopic cracks. The interplay between bone stiffness, collagen crosslinking and fluid pressurization dictates whether damage remains subclinical or evolves into a stress fracture.
Imaging and Quantification Techniques
High-resolution tools allow researchers to map the spatial distribution and propagation of microcracks. Ex vivo assessments use scanning electron microscopy (SEM) to visualize crack morphology. Confocal laser scanning microscopy can highlight damaged regions after staining with fluorescent dyes. In living subjects, advanced in vivo imaging modalities—such as microcomputed tomography—enable longitudinal tracking of crack initiation and repair, bridging experimental findings to clinical scenarios.
Cellular and Molecular Response to Microdamage
Early Signaling Events
Within hours of crack formation, nearby osteocytes sense mechanical perturbations through deformation of their lacuno-canalicular network. These cells release cytokines and signaling molecules that orchestrate programmed cell death, or apoptosis, in the most damaged regions. Apoptotic osteocytes mark the precise sites where remodeling should commence, while surviving osteocytes propagate distress signals to recruit effector cells.
Recruitment of Remodeling Units
- Osteoclastic Phase: Mononuclear precursors fuse to form active osteoclasts that focus on resorbing the cracked bone surface, creating a resorption pit called the Howship’s lacuna.
- Reversal Phase: Mononuclear cells prepare the pit floor by secreting matrix proteins and growth factors, setting the stage for new bone formation.
- Formative Phase: Osteoblasts populate the resorption cavity, secreting osteoid that will later mineralize and fill the defect.
Bone Remodeling and Repair Mechanism
Integration of Mechanical and Biological Factors
The field of mechanobiology explores how mechanical cues influence cellular functions during repair. Strain gradients around a crack modulate gene expression in osteogenic cells, promoting localized proliferation and matrix deposition. Concurrently, angiogenic factors stimulate new vessel ingrowth to supply nutrients and remove waste, ensuring effective healing.
Phases of Tissue Replacement and Mineralization
Once osteoid is laid down, a controlled cascade of mineral nucleation begins. Calcium phosphate crystals infiltrate the organic scaffold, restoring stiffness and strength. Over weeks to months, the remodeling cycle gradually replaces older, damaged bone with mature lamellar tissue. The final architecture often mirrors the original microstructure, preserving overall bone resilience.
Clinical Implications and Future Directions
Recognizing the dynamics of microcrack repair informs strategies to prevent stress fractures in athletes and elderly patients alike. Pharmacological agents may target osteocyte signaling pathways to enhance targeted remodeling, while biomaterials research focuses on scaffolds that mimic the natural bone environment. Biomechanical conditioning programs aim to optimize loading patterns that maintain a balanced turnover, reducing the risk of overuse injuries. Continued integration of high-resolution imaging, molecular biology and computational modeling promises to unveil novel therapeutics that safeguard skeletal health throughout life.