The integration of bone implants within the human body demands a delicate balance between mechanical strength and immunological acceptance. Successful implantation relies not only on the **biomechanical** compatibility of prosthetic devices but also on their capacity to evoke a controlled **immune** response that fosters tissue regeneration rather than chronic inflammation. This article explores the interplay between **biomaterials**, host defense mechanisms, and cutting-edge strategies aimed at enhancing **osseointegration** while minimizing adverse reactions.
Biological and Material Considerations
Bone replacement and augmentation procedures frequently utilize a variety of **bioceramics**, metals, and polymer composites. Materials such as titanium alloys and hydroxyapatite offer high **biocompatibility** and resistance to corrosion, but each possesses unique challenges in modulating the host response.
Material Selection
- Titanium: Favored for its mechanical strength and inert behavior; surface treatments can improve cellular adhesion.
- Ceramics: Hydroxyapatite and calcium phosphates mimic the mineral phase of bone, enhancing osteoinductive potential.
- Polymers: Biodegradable polymers like polylactic acid enable controlled degradation, but may generate acidic byproducts.
Surface Properties
Surface topography and chemistry are critical in directing cellular responses. Roughened or nanopatterned surfaces increase contact area, promoting **osteoblast** attachment and differentiation. Chemical modifications, such as immobilizing peptides or growth factors, can further tailor the implant’s **bioactivity**.
Immunological Interactions at the Implant Site
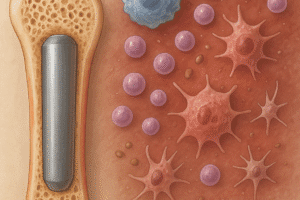
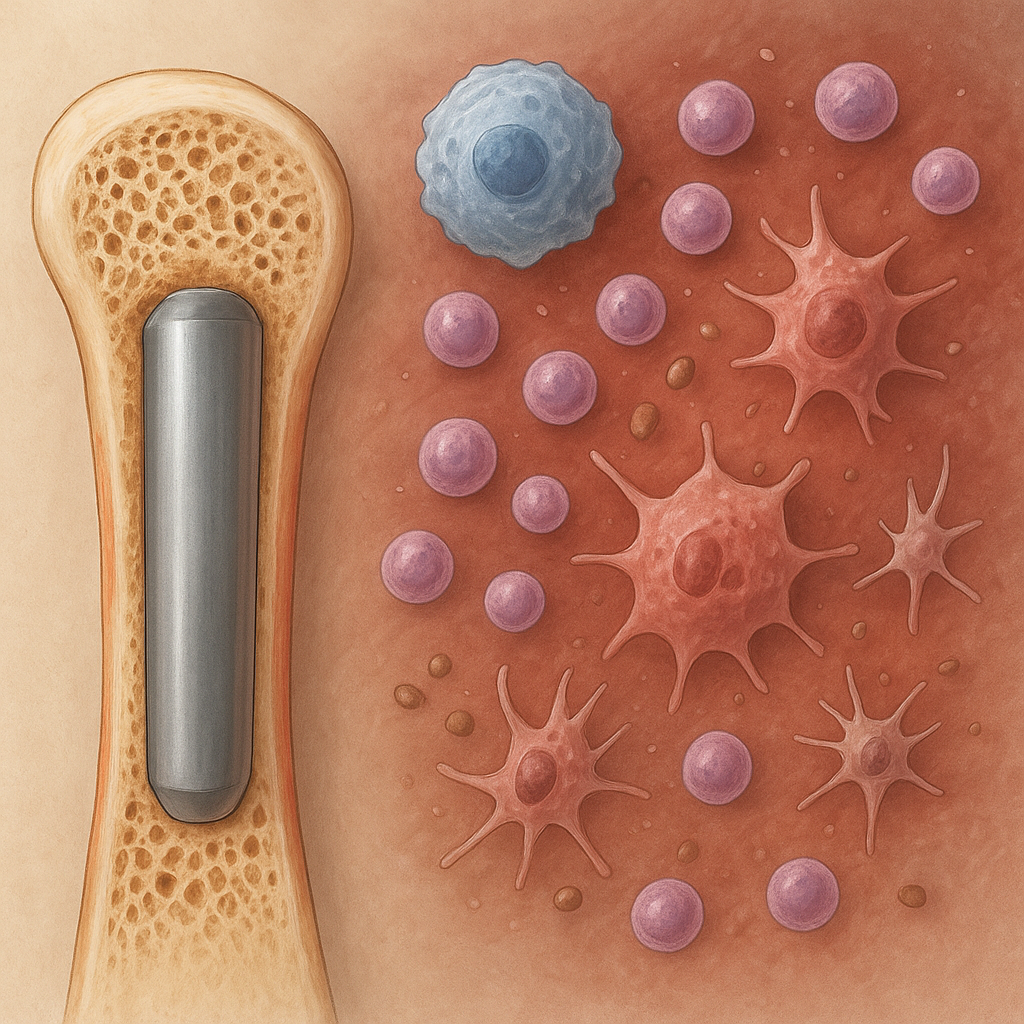
Upon implantation, the body mounts an innate immune response that can dictate long-term success or failure. The initial phase involves protein adsorption on the implant surface, followed by recruitment of **neutrophils** and **macrophages**. The polarization state of macrophages (M1 pro-inflammatory versus M2 pro-regenerative) plays a pivotal role in healing outcomes.
Innate Immunity and Foreign Body Response
- Protein Corona: Adsorbed plasma proteins such as fibrinogen and albumin form a provisional matrix that influences cell adhesion.
- Macrophage Activation: M1 macrophages release pro-inflammatory **cytokines** (e.g., TNF-α, IL-1β), whereas M2 macrophages secrete anti-inflammatory mediators (e.g., IL-10) that support tissue repair.
- Giant Cell Formation: Persistent irritants can lead to foreign body giant cells and fibrous encapsulation, isolating the implant.
Adaptive Immunity
T cells and B cells may also participate in chronic responses. Surface-bound antigens or leached degradation products can act as haptens, triggering specific lymphocyte activation. Balancing **adaptive immunity** is crucial to prevent hypersensitivity or implant rejection.
Advanced Biomaterials and Immunomodulation
Innovations in material science aim to steer the immune environment toward regeneration. Strategies include functionalizing surfaces with bioactive molecules, engineering **nanostructures**, and delivering immunomodulatory agents locally.
Surface Functionalization
- Peptide Grafting: RGD sequences and other adhesion motifs encourage **osteoprogenitor** cell binding and proliferation.
- Growth Factor Release: Gradual elution of BMP-2 or VEGF supports angiogenesis and bone formation.
- Antimicrobial Coatings: Silver nanoparticles or antibiotic-loaded polymers reduce infection risk without overly suppressing host immunity.
Nanotechnology and Controlled Release
Nanoscale features and carriers such as liposomes or polymeric nanoparticles enable precise delivery of anti-inflammatory cytokines (e.g., IL-4) or siRNA to modulate gene expression in situ. These approaches aim to shift macrophage polarization toward a pro-healing phenotype and minimize fibrotic encapsulation.
Clinical Implications and Future Perspectives
Despite remarkable progress, challenges remain in translating laboratory successes into routine clinical practice. Long-term **clinical trials** are essential to assess implant durability, host response, and functional integration under load-bearing conditions.
Infection and Implant Failure
- Periprosthetic infection remains a leading cause of revision surgeries; early detection and **antimicrobial** strategies are vital for patient outcomes.
- Aseptic loosening often arises from chronic inflammation and osteolysis mediated by wear particles and immune activation.
Personalized Implants
Emerging trends include patient-specific implants designed via 3D printing, incorporating **surface modification** tailored to individual immunoprofiles. Genomic and proteomic diagnostics may guide the selection of immunomodulatory coatings or systemic therapies to optimize implant acceptance.
Regenerative Medicine Integration
Combining **stem cell** therapies with immunologically informed scaffolds holds promise for true bone regeneration, surpassing simple prosthetic replacement. Future research into the crosstalk between immune cells and osteogenic lineages will refine these approaches, ultimately enhancing patient quality of life.