Bone healing involves a complex interplay of cellular events and biomechanical factors that vary significantly between pediatric and adult patients. Understanding these differences is essential for optimizing treatment strategies, improving recovery times, and minimizing long-term complications. This article explores the unique aspects of fracture repair in children and adults, highlighting physiological variations, clinical considerations, and emerging research trends.
Growth Plate and Remodeling Dynamics
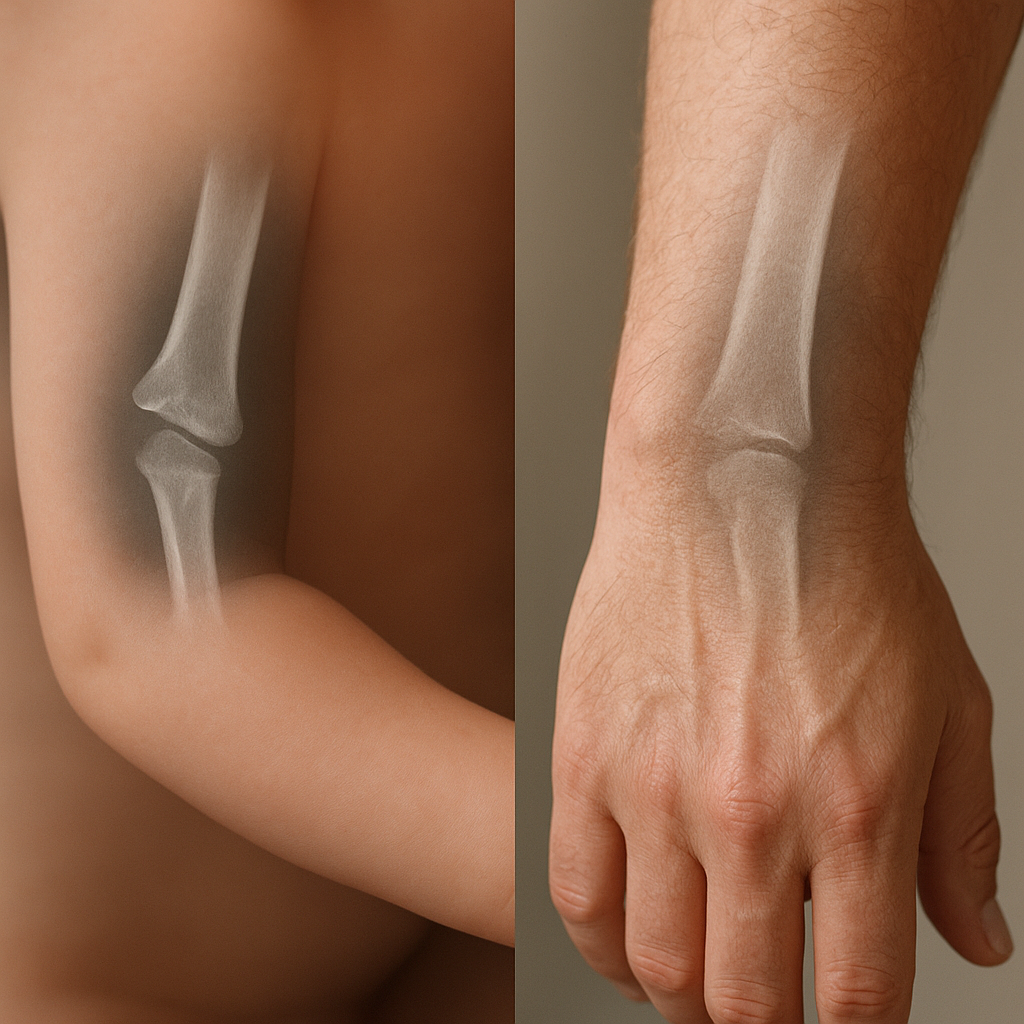
The presence of active growth plates, or physes, in pediatric patients introduces a distinct environment for osteogenesis. In children, the physis serves as a highly vascularized zone of proliferating chondrocytes that enables rapid longitudinal bone growth. This region contributes not only to lengthening but also to efficient remodeling of microdamage and malunited fragments.
Physis Structure and Function
The growth plate consists of four zones—reserve, proliferative, hypertrophic, and calcification—each orchestrating stages of cartilage differentiation and mineral deposition. Damage to the physis can lead to angular deformities or growth arrest, making surgical intervention and fixation methods uniquely challenging. The periosteum in children is thicker and richer in stem cell niches, which accelerates callus formation and supports the bridging of fracture gaps.
Remodeling Capacity
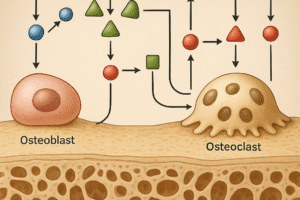
Children exhibit an exceptional ability for spontaneous correction of deformities through continued growth. Even fractures with residual angulation often remodel over time, leveraging the high osteoblastic and chondrogenic activity. The process is guided by mechanical stresses and growth signals, resulting in nearly complete restoration of bone geometry. Conversely, adults have reduced periosteal activity, and remodeling relies primarily on targeted bone turnover driven by osteoclasts and osteoblasts at the fracture site.
Physiological Differences in Fracture Repair
Key physiological processes such as inflammation, vascularization, and cell recruitment differ with age. These variations affect the timeline and quality of bone healing, influencing clinical decision-making.
Inflammatory Phase
Immediately after injury, a hematoma forms, releasing cytokines and growth factors that recruit inflammatory cells. In pediatric patients, the inflammatory response is often more controlled, with rapid transition to the repair phase. Adults, especially those with comorbidities like diabetes or smoking history, may experience prolonged inflammation, increasing the risk of nonunion.
Repair Phase and Callus Formation
The repair phase is characterized by callus formation, involving mesenchymal stem cell differentiation into chondrocytes and osteoblasts. Children generate a robust external callus quickly, thanks to abundant stem cells in the periosteum and bone marrow. Adults produce callus more slowly, and excessive motion or inadequate stabilization can result in delayed union or hypertrophic nonunion.
- Children: Rapid bridging callus, high cellularity, quick transition to remodeling.
- Adults: Thicker lamellar bone, dependence on internal fixation, slower epithelialization.
Remodeling Phase
Following callus maturation, the remodeling phase restores the bone’s original shape and mechanical strength. Pediatric bones remodel under the influence of growth signals and mechanical loads, often achieving complete anatomical restoration. In adults, however, remodeling is a longer, less dynamic process that may require interventions such as bone grafting or pharmacological agents to enhance angiogenesis and matrix synthesis.
Clinical Implications of Age-Related Differences
Treatment strategies must be tailored to leverage the inherent strengths of each age group while mitigating risks associated with physiological limitations. Considerations include fixation devices, weight-bearing protocols, and rehabilitation plans.
Fixation Techniques
In pediatric fractures, flexible intramedullary nails or elastic stable intramedullary nailing (ESIN) systems are favored, accommodating growth and allowing micro-motion to stimulate healing. Plating is reserved for complex or open fractures where anatomical reduction is critical. For adults, locking plates and rigid intramedullary nails provide the biomechanical stability needed to compensate for slower bone regeneration.
Weight-Bearing and Rehabilitation
Children often progress to full weight-bearing sooner due to rapid callus strength acquisition. Physiotherapists focus on restoring range of motion and normal gait patterns quickly. Adults may require protected weight-bearing for extended periods, with gradual load progression to avoid hardware stress and promote adequate callus maturation.
Risk of Complications
Key complications differ by age group:
- Pediatric risks: Growth arrest, physeal bar formation, and angular deformity.
- Adult risks: Delayed union, nonunion, and post-traumatic osteoarthritis.
Early detection of complications and timely revision surgery—such as osteotomy or bone grafting—are crucial to achieving positive outcomes.
Innovations and Research Directions
Advances in biotechnology and biomaterials are shaping the future of fracture management across age groups. Emerging therapies aim to harness molecular pathways and improve clinical outcomes.
Biological Enhancers
Recombinant growth factors, such as BMP-2 and BMP-7, have shown promise in enhancing bone repair, particularly in adults with compromised healing capacity. Pediatric applications require caution due to potential effects on growth plates. Research is ongoing to develop targeted delivery systems that maximize local efficacy while minimizing systemic exposure.
Scaffold Technologies
Three-dimensional printed scaffolds infused with mesenchymal stem cells and osteoinductive coatings offer customizable solutions for segmental defects. These constructs aim to replicate native bone architecture and support vascular ingrowth. Preclinical models demonstrate accelerated union in both juvenile and mature bone environments.
Gene Therapy and Cellular Approaches
Gene editing techniques targeting signaling molecules like VEGF and PDGF hold the potential to modulate the local microenvironment and boost vascularization. Additionally, autologous stem cell transplantation is under investigation to directly repopulate fracture sites with osteoprogenitor cells, potentially reducing reliance on hardware and external fixation.
Personalized Treatment Planning
Precision medicine integrates patient-specific factors, including age, bone quality, genetic predispositions, and lifestyle, to tailor fracture management protocols. Computational modeling and finite element analysis assist surgeons in evaluating fixation strategies under various loading scenarios. This personalized approach aims to optimize healing time, reduce complications, and enhance functional recovery across both pediatric and adult populations.