The following article examines recent achievements in the field of spinal fusion and bone growth enhancement, emphasizing both well‐established methods and pioneering therapies. By exploring cellular mechanisms, technological innovations, and clinical applications, readers will gain insight into how these advances are reshaping outcomes for individuals with spinal disorders and injuries.
Cellular and Biological Foundations of Fusion
Successful fusion relies on coordinated events that initiate and sustain osteogenesis. At the core is the recruitment of mesenchymal stem cells to the site of injury, where they differentiate into osteoblasts under the influence of growth factors and mechanical cues. Traditional approaches use autologous bone graft to provide an osteoconductive scaffold, osteoinductive signals, and living cells. However, donor‐site morbidity and limited graft volume have inspired the development of synthetic and biologic alternatives.
Growth Factor Strategies
Recombinant proteins such as BMP (bone morphogenetic proteins) have demonstrated potent osteoinductive activity. BMP‐2 and BMP‐7 are approved for selected spinal fusion procedures, accelerating new bone formation by upregulating osteoblast gene expression. Researchers are also investigating novel cytokines and small molecules to modulate the inflammatory phase, optimize angiogenesis, and fine‐tune the balance between bone resorption and formation.
Scaffold and Material Innovations
Biodegradable polymers, ceramics, and composite materials now serve as tunable scaffolds. Advances include nanostructured surfaces that mimic the extracellular matrix, promoting cell adhesion and proliferation. Porosity gradients facilitate vascular ingrowth, while controlled degradation rates match the pace of new bone formation. Osteoconductive hydroxyapatite and bioactive glass are often combined with collagen or polycaprolactone to yield mechanically stable, resorbable implants.
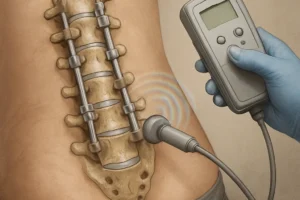
Mechanical and Electrical Bone Growth Stimulation
In addition to biologics, physical stimuli can enhance fusion. Mechanical stimulation through controlled micromotion and compression encourages bone remodeling according to Wolff’s law. Novel devices apply axial loads or torsional oscillations to spinal segments, activating mechanotransduction pathways in osteoblasts and osteocytes.
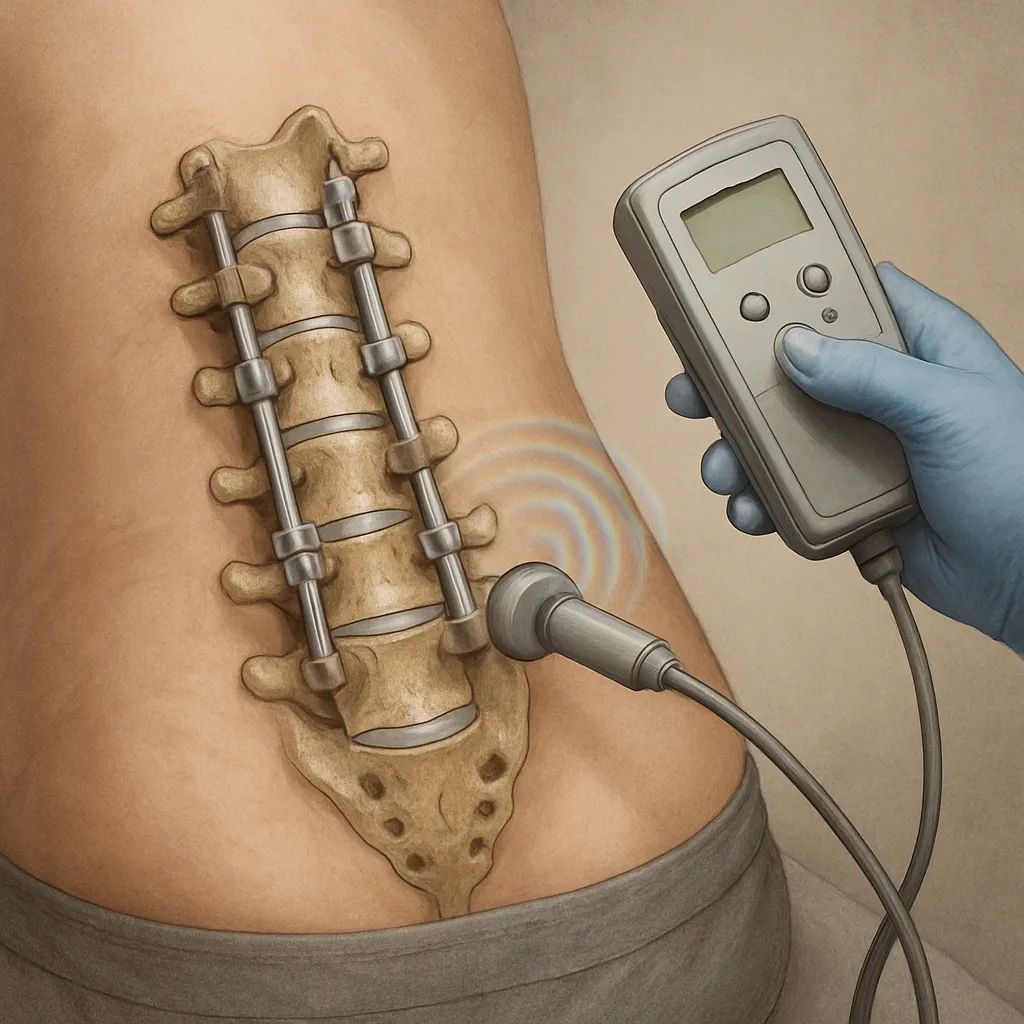
Electric and Electromagnetic Modalities
Low‐intensity pulsed ultrasound (LIPUS) and pulsed electromagnetic fields (PEMF) have a growing evidence base. These non‐invasive methods alter ionic flow across cell membranes, upregulating genes associated with matrix synthesis. Clinical trials report reduced time to fusion and lower rates of pseudoarthrosis when PEMF is used adjunctively with grafts or cages.
- Non‐invasive delivery, patient‐controlled
- Minimal side effects
- Compatibility with minimally invasive approaches
Implantable Mechanical Devices
Dynamic interbody spacers and expandable cages provide sustained distraction and optimize load sharing. Some designs incorporate springs or shape‐memory alloys that adjust height post‐implantation. These implants maintain proper alignment, preserve foraminal dimensions, and promote a microenvironment conducive to bone deposition.
Advances in Surgical Techniques and Instrumentation
Modern spinal fusion benefits from refined surgical approaches that reduce tissue trauma and enhance precision. Endoscopic and navigation‐assisted systems permit minimally invasive access, smaller incisions, and faster recovery. Intraoperative imaging and robotics further improve accuracy when placing pedicle screws and interbody devices, reducing complications related to malposition.
Enhanced Visualization and Navigation
Three‐dimensional fluoroscopy and O‐arm imaging integrate seamlessly with neuronavigation platforms. Surgeons visualize screw trajectory in real time, ensuring optimal purchase in osteoporotic bone. Augmented reality overlays and haptic feedback tools are emerging, helping to maintain safety margins around neural elements.
Hybrid Fixation Systems
Combining dynamic rods with rigid screws yields hybrid constructs that balance immediate stability with controlled micromotion. This design can reduce adjacent segment degeneration by more naturally transferring loads. Instrumentation manufacturers now offer pre‐contoured implants and modular connectors to streamline intraoperative assembly.
Clinical Outcomes and Patient‐Centered Considerations
Advances in fusion and bone stimulation translate to measurable improvements in patient outcomes. Reduced operative time, lower blood loss, and expedited mobilization contribute to faster discharge and improved quality of life. Radiographic fusion rates have climbed above 90% in many centers, even in challenging revisions and deformity corrections.
Risk Stratification and Personalized Medicine
Precision medicine tools, such as genetic profiling and biomarkers of bone turnover, enable individualized treatment plans. Patients with risk factors—osteoporosis, smoking, diabetes—may receive tailored regimens combining high‐dose BMP, local drug delivery of anti‐resorptives, and customized rehabilitation protocols. Software models predict biomechanical outcomes, assisting surgeons in selecting construct stiffness and graft composition.
Rehabilitation and Functional Recovery
Early mobilization protocols integrate physical therapy with adjunctive bone stimulation devices. Wearable sensors monitor gait, posture, and spinal alignment, providing feedback for therapists. Virtual reality–based exercises engage patients in targeted muscle strengthening while avoiding undue stress on the fusion site.
Emerging Horizons in Regenerative Spine Care
Next‐generation strategies aim to go beyond fusion, exploring true disc regeneration and segmental motion preservation. Tissue engineering approaches deliver stem cells within 3D‐printed scaffolds engineered to release growth factors in a spatially controlled manner. Gene therapy vectors targeting Runx2 and Sox9 transcription factors hold promise for in situ reprogramming of resident progenitors.
Bioreactor‐Conditioned Constructs
In vitro cultivation of bone graft substitutes within bioreactors exposes cells to dynamic perfusion and mechanical loading, preconditioning the tissue for implantation. These living grafts exhibit accelerated integration and superior mechanical properties compared to static constructs.
Immunomodulation and Host Integration
Understanding the role of macrophage polarization has led to materials that actively recruit M2 phenotype cells, minimizing inflammation and promoting healing. Surface coatings with anti‐inflammatory peptides or controlled release of IL‐10 can steer the immune response toward a tissue‐regenerative profile.
Collectively, these advances represent a paradigm shift in spine surgery, moving from purely structural repair toward biologically integrated solutions. By harnessing the synergy of biology, mechanics, and technology, the field is poised to deliver safer, more effective treatments for patients with degenerative, traumatic, and congenital spinal conditions.