The integration of engineered materials with the body’s natural healing processes has transformed the landscape of orthopaedic surgery. Evaluating clinical outcomes in bone reconstruction requires a multi-dimensional approach that addresses not only structural repair but also restoration of biological and functional integrity. This article explores the latest strategies for assessing bone healing, the role of advanced biomaterials, and the key clinical metrics that define success in reconstructive procedures.
Methodologies for Assessing Bone Reconstruction
A rigorous evaluation framework combines imaging, mechanical testing, and biological assays. These techniques guide surgeons and researchers in determining the degree of osteointegration and overall graft performance.
Radiological and Imaging Techniques
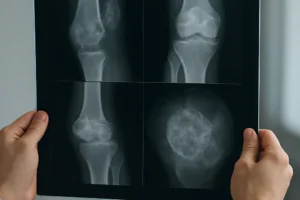
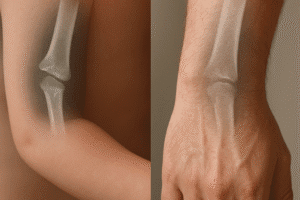
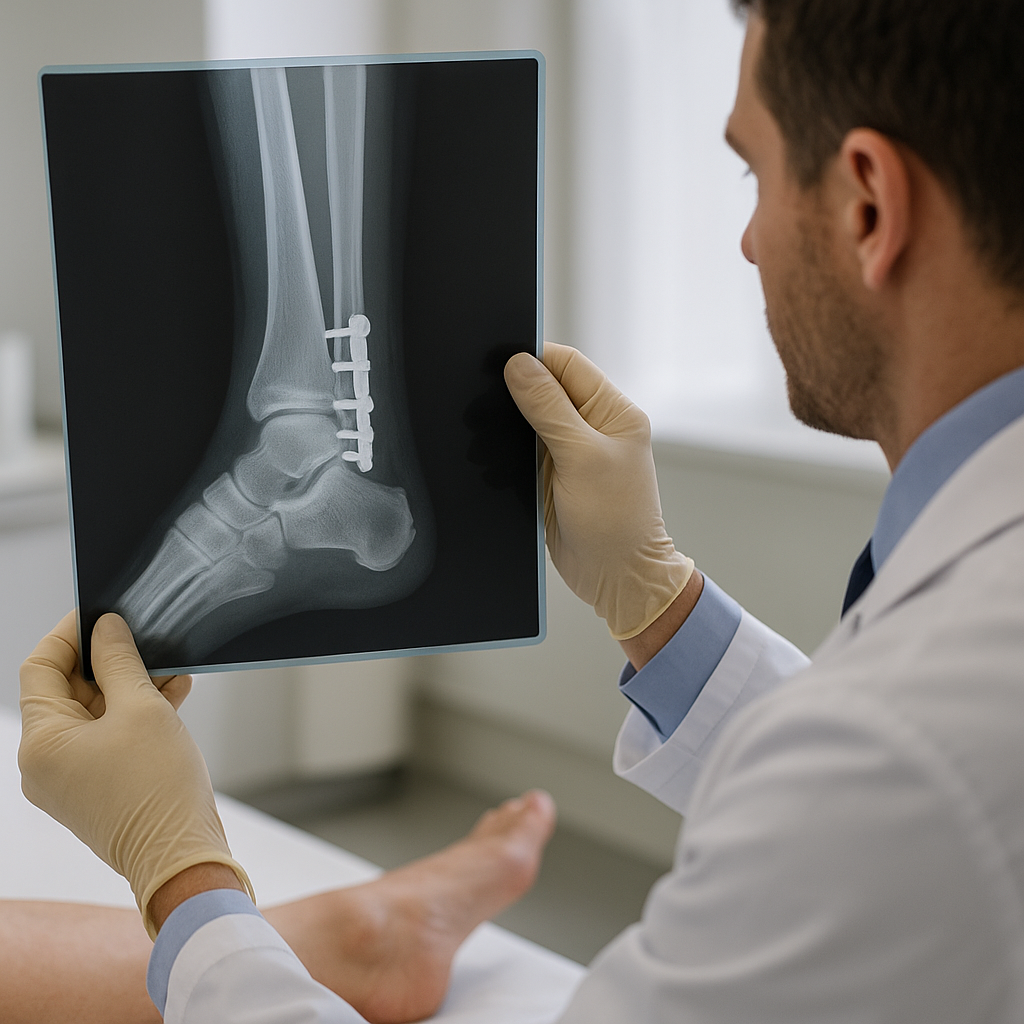
- X-ray monitoring provides a quick snapshot of callus formation and implant positioning but has limited sensitivity for early cellular events.
- Computed Tomography (CT) enables precise volumetric measurements of bone density and can detect subtle resorption or graft failures.
- Magnetic Resonance Imaging (MRI) is invaluable for visualizing soft tissue–bone interfaces and assessing postoperative vascularization.
- Emerging modalities such as dual-energy CT and PET/CT enhance detection of metabolic activity, reflecting ongoing regeneration.
Biomechanical and Functional Testing
Beyond imaging, evaluating the mechanical competence of a reconstructed bone is essential. Load-to-failure tests, axial compression assessments, and torsional stiffness measurements reveal whether the repair can withstand physiological forces.
- In vitro mechanical tests on explanted specimens validate the strength of scaffold constructs.
- Gait analysis and force plate studies in animal models simulate real-world functional demands, highlighting discrepancies between structural integration and mechanical performance.
- In vivo load sensors embedded in implants allow continuous monitoring of mechanical loading patterns during patient rehabilitation.
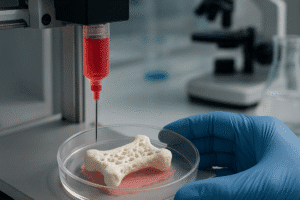
Advanced Biomaterials and Technologies
The choice of biomaterial significantly influences the outcome of bone reconstruction. Optimal scaffolds must balance biocompatibility, mechanical strength, and resorption kinetics to support new bone formation.
Ceramic-based Scaffolds
- Hydroxyapatite and tricalcium phosphate closely mimic the mineral component of bone, promoting direct bonding to host tissue.
- Porosity and interconnectivity are tailored to facilitate cellular migration and angiogenesis.
- Surface modifications with bioactive peptides or growth factors accelerate early bone deposition.
Polymeric and Composite Materials
Biodegradable polymers such as polylactic acid (PLA) and polycaprolactone (PCL) offer customizable degradation rates. When reinforced with ceramic particles or carbon fibers, they achieve enhanced mechanical performance while remaining resorbable.
- Electrospun nanofibers create a biomimetic extracellular matrix for osteoblast adhesion.
- Injectable hydrogel composites allow functional filling of irregular defects and can deliver therapeutic agents on demand.
- 3D printing techniques produce patient-specific constructs with precise anatomical fit and controlled porosity gradients.
Biological Adjuncts and Cell-based Therapies
Incorporating cells and biologics into bone graft substitutes enhances healing potential. Mesenchymal stem cells (MSCs) secrete osteoinductive factors and differentiate into bone-forming lineages under appropriate stimuli.
- Platelet-rich plasma (PRP) delivers autologous growth factors that modulate inflammation and stimulate regeneration.
- Gene-activated matrices encode for bone morphogenetic proteins (BMPs) to establish a localized production of osteogenic signals.
- Co-culture systems combining endothelial cells with osteoprogenitors improve early vascular network formation within the scaffold.
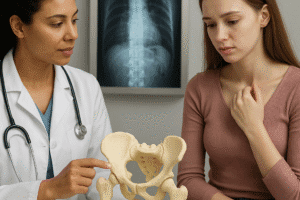
Clinical and Functional Outcomes
Translating laboratory advances into reliable clinical solutions demands thorough tracking of both short-term safety and long-term functionality. The following parameters help define the effectiveness of bone reconstruction therapies.
Patient-reported Outcome Measures (PROMs)
- Pain scores, mobility scales, and quality-of-life questionnaires capture the subjective experience of patients and guide postoperative care.
- Standardized tools such as the Visual Analog Scale (VAS) and the Short Form 36 (SF-36) facilitate comparative analyses across treatment modalities.
Objective Functional Tests
Objective assessments complement PROMs by quantifying functional gains:
- Range of motion (ROM) measurements evaluate joint flexibility adjacent to the reconstruction site.
- Timed up and go (TUG) and six-minute walk tests (6MWT) assess overall mobility and endurance.
- Biomechanical gait analysis profiles loading symmetry and compensatory mechanisms that may affect long-term outcomes.
Long-term Surveillance and Complication Rates
Monitoring complications such as infection, implant loosening, or nonunion is critical:
- Periodic radiographic follow-ups detect early signs of implant failure or graft resorption.
- Blood and tissue markers of inflammation guide interventions before clinical symptoms arise.
- Registries and multicenter trials provide real-world evidence on the durability and safety of novel reconstruction strategies.
Key considerations include patient comorbidities, defect size, and host biology, all of which interplay with the chosen biomaterial and surgical technique. A multidisciplinary team approach—combining orthopaedic surgeons, biomedical engineers, and rehabilitation specialists—maximizes the probability of durable, functional bone healing.